Development of Effective Therapies against Liver Fibrosis
Global Journal of Pathology & Laboratory Medicine
Volume 1, Issue 4, December 2021, Pages: 1-50
Received: September 01, 2021; Reviewed: September 04, 2021; Accepted: September 13, 2021; Published: December 30, 2021
Unified Citation Journals, Pathology 2021, 1(4) 1-50; https://doi.org/10.52402/Pathology209
ISSN 2754-0952
Authors: Dr. Laila Alshammari
Master of Clinical Sciences (MSc), Bangor University
B.Sc Medical Laboratory (2014, Northern Border)
Keywords: Hepatic fibrosis, antifibrotic, hepatitis, cirrhosis, chronic liver disease
1. Abstract:
Liver fibrosis commonly occurs as a liver’s reaction to a persistent injury. Some of the significant causes of LF include hepatitis and alcohol ingestion. The primary intention of this exploration was to deduce the development of the potential therapeutic agents that can be effective in managing patients diagnosed with hepatic fibrosis. The exploration involved a systematic qualitative review of one hundred and twenty articles outsourced from five major databases, namely, Web of Science, Science Direct, PubMed, Cochrane Library, and the BMC. A review of the articles aimed at unveiling the drugs that are potent in treating hepatic fibrosis in humans. The outcomes of the investigation revealed that even though many studies exist on some of the drugs that show antifibrotic effects, few trials involved human models. As such, there is a need to carry out more explorations on the agents that demonstrate antifibrotic potential on animal models. The most operational way of managing patients who present with hepatic fibrosis is treating the triggering factor, such as giving antivirals for viral hepatitis.
1. Introduction:
The section will cover some of the critical areas related to the diagnosis and, most imperatively, the management of the condition as per the existing explorations. The chapter will aim at providing some essential background highlights on the treatment of liver fibrosis while offering some objectives of the study, which, in a way, relate to the research questions. The primary intention of this chapter will be to outline the various aspects discussed in the following chapters while offering the existing research gap. Following this chapter, which gives a preview of the dissertation, there will be chapter two, which will mainly compose of a literature review. Chapter three will cover the methodology, while the subsequent chapter will contain the results of the study, including the data analysis. The last chapter will be a conclusion covering the specific highlights of the exploration, illustrating the importance of the findings. The last chapter will also include some recommendations in line with the research findings.
1.2 Background
Fibrosis is a response that mainly occurs in the body following a long-lasting injury as a way of trying to invoke normal integrity of the hepatic tissues as necrosis or apoptosis takes place (Schuppan & Kim, 2013). In case the damage persists for a long time, the fibrosis often culminates into the formation of scar tissue in hepatocytes as it happens in liver cirrhosis. Antifibrotic therapies form the most promising route of enhancing the management of hepatic fibrosis even though high risks exist with the use of therapeutic agents. Most preclinical explorations yield several agents that can act as targets for the antifibrotic agents. However, most of such explorations stopped because of the lack of biomarkers that have high sensitivity and specificity to determine either the progress or the reversal of hepatic fibrosis (Schuppan & Kim, 2013). As such, there is a need to unveil some of the concrete pathways resulting in liver fibrosis and subsequently to work towards developing effective therapies.
Over the past years, numerous investigators have taken part in exploring effective therapies for LF. Generating a proper comprehension of the molecular mechanisms that enhances the process of hepatic fibrosis has been among the widely researched areas when trying to develop novel models of managing the condition when not focusing only on the etiologic agents (Trautwein et al., 2015). Even though researchers show the antifibrotic activity of some of the agents in animal models, none of the compounds has proper validation for use among humans in clinics or as an effective therapy for hepatic fibrosis. As such, there is a possibility that researchers need to develop combination therapies that can effectively manage liver fibrosis (Trautwein et al., 2015). Research is the best way to uncover the best combination of therapies for hepatic fibrosis.
1.3 Research Aims and Objectives
1.3.1 Research Aims
The purpose of the exploration is:
- To evaluate the prevailing data on the management of liver fibrosis.
- To enhance the existing knowledge on effective therapies for liver fibrosis.
1.3.2 Research Objectives
The chief intentions of this investigation will be:
- To depict an in-depth understanding and review of peer-reviewed materials related to the topic.
- To critically review and synthesize prevailing explorations on the treatment of liver fibrosis.
- To effectively come up with an accurate strategy for managing patients who develop hepatic fibrosis.
1.4 Rationale
Given that there is a rise in the number of people developing liver cirrhosis globally, there is a need to develop a strategy of utilizing various therapies to manage the condition effectively.
Research shows that even though the mortality of alcohol-related hepatic disease is on the rise despite having stagnated for almost three decades (Julien et al., 2020, p. e316). Most mortality cases occur secondary to liver fibrosis and its complications. As such, there is a need to recognize the best practical strategies for treating liver fibrosis..
1.5 Conclusion
Chapter one provides an excellent description of the primary theme of this exploration. It offers a background of the central theme enabling the reader to have a clear understanding of the primary theme under research and the existing gaps. The chapter also provides the aims, objectives, and rationale of the study, which aligns with the theme. As described in this chapter, the research will follow the systematic approach to present the various aspects of the study in a straightforward way.
Chapter 2: Literature Review
2.1 Introduction
LRs are quite essential for investigators since they often provide to the reader some of the background and up-to-date data concerning the primary theme of the paper. The LRs offer a wide range of data to the extent that a researcher can recognize the prevailing research gap, enabling them to explore such areas (Wee & Banister, 2016). When developing LRs, individuals have to acquire data on a specific topic, which will support the central hypothesis of the research. Such data is easily accessible via various online search engines such as Google (Winchester & Salji, 2016). However, the investigators have to utilize peer-reviewed materials that contain well-researched and accurate information related to various topics. LRs have the role of bringing up the context and current thinking while not necessarily offering a specific question (Robinson & Lowe, 2015). As such, LRs offer a general picture of the topic using the existing data.
This chapter is inclusive of an introduction, main section, and a conclusion that offers some original conclusions of the findings under the literature review section. Under the main section, there will be a discussion of the primary literature strategy and a critique of various peer-reviewed materials which contain relevant information in line with the topic. The main section will also include a section that offers some critical background data on the pathogenesis of LF, which is decisive in understanding the evolution of various drugs for the management of the condition.
2.2. Literature Search Strategy
2.2.1 Informational sources
When carrying out a systematic literature review, the researcher must make it clear how they identify the various studies used in the exploration (Cooper et al., 2018).
Consequently, there was a search for the primary themes leading to the analysis of both primary and secondary sources. When working on finding the sources, there was a primary focus on selecting only peer-reviewed materials. As such, most of the sources were from credible sources such as Google Scholar and PubMed. Another significant inclusion criteria when selecting the sources was the year of publication. The dissertation only utilizes data from articles published within the past ten years, which ensures the accuracy and credibility of the data.
2.2.2 Focused searches
When doing the searches to develop a list of sources that could be useful when working on this dissertation, five databases provided the primary sources, as depicted in Table 2 below. The top search terms utilized during the process of looking for credible journal articles were “effective therapies”, “management”, “liver fibrosis,” and “pathogenesis”. Besides, there was a combination of the search terms with keywords such as “for” and “of” to ensure that the search would provide only articles that focused on the primary theme. The articles chosen had to be in the English language, and their date of publication was in the past ten years. Another filtering option when searching was the availability of the whole article online. Only articles that were fully accessible online without the need for subscriptions were taken into account. The initial search in the five databases revealed that 89,215 articles contained data related to the theme of the dissertation. However, following the application of the various filters only left only 16,595 articles that were viable for analysis during the study, even though some were not entirely relevant. Out of this number of journal articles, there was the selection of the sources that provided detailed data concerning the topic quickly for the reader to easily understand, enabling the consequent analysis of such articles to be comfortable when participating in the systematic review.
Table 2.1: The literature searching strategy
2.3 Main Themes in Literature
Following an in-depth analysis of a vast range of peer-reviewed materials from the five databases, there were identifications of various themes that play an excellent platform for understanding the topic of this dissertation. The themes are in line with the research topic and would play a noteworthy part in enabling the reader to fathom the effective treatment of liver fibrosis. As such, it is imperative to analyze all the themes in a detailed manner. The themes are inclusive of:
- Introduction to liver fibrosis (Petitclerc et al., 2017)
- Epidemiology of liver fibrosis (Tapper &Parikh, 2018)
- Pathogenesis of LF (Altamirano-Barrera, et al., 2017)
- Diagnosis of LF (Lurie et al., 2015)
- Therapeutic agents of hepatic fibrosis. (Schuppan & Kim, 2013)
2.3.1 Introduction to liver fibrosis
LF (Liver Fibrosis) mainly signifies that a patient has had chronic liver disease (CLD). The main features include the build-up of the excess quantities of extracellular (EC) matrix proteins (Petitclerc et al., 2017). In case a patient who has LF does not acquire timely and active treatment, the fibrosis often progresses to cirrhosis, which acts as one of the major predisposing factors for hepatocellular carcinoma (HCC) (Forner et al., 2012). As such, there is a requisite to advance effective remedies to avert liver disease progression to cirrhosis.
2.3.2 Epidemiology of liver fibrosis
CLD and cirrhosis accounted for more than one million deaths globally in the year 2015 (Wang et al., 2016). Studies show that fibrosis resulting in cirrhosis and HCC often culminate into a financial and social burden to not only the patient and their family members but also the entire society (Bajaj et al., 2011; Patel et al., 2017; Tapper et al., 2015). It is quite demoralizing to note that the rates of cirrhosis have doubled in the past ten years in America (Beste et al., 2015). The trend is likely to endure despite the widespread advancements in the production of effective antiviral regimens that target Hepatitis C, which is among the chief contributors to cirrhosis (Moon et al., 2017). In line with this, mortalities occurring secondary to cirrhosis complications are likely to triple in the next ten years (Estes et al., 2018). The main driving factors of such an anticipated increase in cases of cirrhosis include the rise in alcohol liver illnesses and NASH (Mellinger et al., 2018; Parikh et al., 2019; Younossi et al., 2011). However, it is fundamental to note that the prevalence and outcomes of liver cirrhosis vary widely in America (White et al., 2017). Studies indicate that prevention and early diagnosis followed by treatment of cirrhosis reduces the overall cost of acquiring quality healthcare (Rein et al., 2012; Tapper et al., 2015; Tapper et al., 2016). Thus, effective treatment agents for LF are essential to reduce the prevalence of cirrhosis and HCC.
2.3.3 Pathogenesis of LF
LF mainly occurs as a product of the pathological deposition of the excess matrix in combination with chronic inflammation (Altamirano-Barrera et al., 2017). LF is prevalent in patients who consume high amounts of alcohol for prolonged periods, those with NAFLD, and hepatitis (Sánchez-Valle et al., 2012).

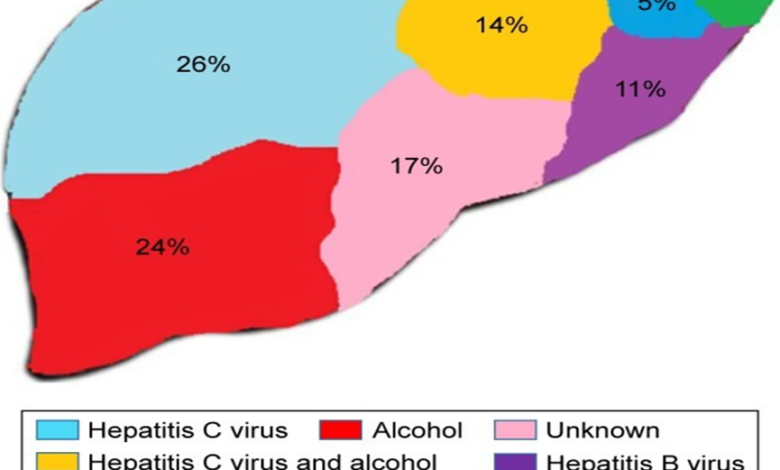
Figure 2.1: Some of the chief contributors of CLD. Source: (Poilil Surendran et al., 2017
 Figure 2.2: Mechanisms leading to LF. Source: (Altamirano-Barrera et al., 2017).
Figure 2.2: Mechanisms leading to LF. Source: (Altamirano-Barrera et al., 2017).
Figures 2.1 and 2.2 above show the causes of LF and how it develops. The initial step following the exposure of the hepatocytes to the various agents is their response. The hepatocytes response plays a noteworthy part in the pathophysiology of LF since it mainly involves the conscription of cells that both hinder and the ones that enhance inflammatory reactions, especially monocytes. Subsequently, there is the assembly of cytokines and other chemokines that act to enhance the recruitment process. The chemokines also stimulate the stellate cells in the liver through activating proinflammatory cells. Among the cytokines released into the bloodstream, TGF-β plays an indispensable part in the pathophysiology of LF. TGF-β enhances the conversion of stellate cells of the hepatic system to myofibroblasts, whose primary role is to secrete the EC matrix enhancing LF (Cinar et al., 2016; Lee et al., 2015). Besides collagen, other EC matrix proteins secreted include fibronectin and laminin (Lee & Friedman, 2011). According to Sebastiani et al. (2014), LF develops as a wound-healing reaction following liver disease, which may either be acute or chronic. It is unfortunate to note that it is not yet certain when fibrosis becomes irreversible. Some of the irreversible changes seen when cirrhosis occurs include portal hypertension and features of liver failure. In case there is early treatment of the cause of fibrosis, such as in Hepatitis C or B infections, reversion often takes place in almost three-quarters of the patients (D’Ambrosio et al., 2012; Marcellin et al., 2013). Such facts call for effective early treatment of LF.
Alcohol intake is among the significant elements that play an influential part in the development of LF. When alcohol undergoes through metabolism, it forms acetaldehyde, and there is a release of ROS (Hernández et al., 2016; Osna et al., 2017; Teschke, 2018). Subsequently, acetaldehyde augments the secretion of TGF-β from the HSC while stimulating the expression of collagen type 1 protein. All these results in hepatic fibrosis. The ROS enhances apoptosis and injury of the hepatocytes (Bian & Ma, 2012; Jeong & Joo, 2016; Redza-Dutord & Averill-Bates, 2016). NASH is another factor that plays a unique part in the advancement of LF. Studies indicate that NASH commonly presents with a raised expression of TGF-β and an upsurge of serum ALT and AST (Poilil Surendran et al., 2017). Collagen deposition is also typical in patients with the condition augmenting LF (Brenner et al., 2012). NASH also augments an upsurge in the levels of FFAs, which ultimately enhances the activation of PPAR-α. Eventually, the process leads to the generation of free ROS that causes apoptosis (Giby & Ajith, 2014). Hepatic fibrosis often results from changes in the primary hepatic cells, including HSC, hepatocytes, Mφ (s), and LSELCs (Bartneck et al., 2014). Once an injury occurs to the liver, Mφ (s) stimulates the fibrosis reaction while activated Kupffer cells rescind the primary hepatic cells while stimulating the activation of HSC (Baeck et al., 2014; Schuppan, 2015; Tanaka & Miyajima, 2016; Zhou et al., 2014). As such, developing compounds that can target fibrolytic features of HSC and Mφ (s) can enhance the development of effective therapies for LF (Li, et al., 2016; Karsdal et al., 2015).
Developing drugs that can target the cells that are inactive (profibrogenic) can work as an effective immunotherapy means of managing hepatic fibrosis (Aravalli & Steer, 2017). Focusing on the pathogenesis of hepatic fibrosis would enable investigators to develop efficient therapeutic agents for managing LF.
The pathogenesis of hepatic fibrosis mainly entails an interaction of processes occurring both at the molecular and cellular levels. Even though there are many causative factors leading to fibrosis in the liver, the pathogenesis involves similar steps (Udompap et al., 2015). Fibrosis in the hepatocytes often forms due to a discrepancy between the assembly and degeneration of the ECM. Among the significant factors that play an essential function in influencing the ECM balance are the levels of matrix metalloproteinases (the enzymes involved in the degeneration of the ECM) and the tissue inhibitors targeting the metalloproteinases (Ebrahimi et al., 2016). The excess amounts of ECM lead to hepatic fibrosis. Hence, effective antifibrotic therapeutic agents would aim at targeting to maintain an equilibrium of ECM levels in the liver.
Numerous cells participate in the establishment of hepatic fibrosis. However, an essential step in the stimulation of the stellate cells in the hepatic system. HSCs reside just next to the sinusoids. Following an injury to the liver, the fibrogenic cytokines released augments the trans-differentiation of the HSCs, which transforms them from the quiescent states to the active state. Among the central cytokines involved in this process include TGF-β, PDGF, and interleukin 1. The activated HSCs express eventually transform into a form that enhances fibrogenesis and chemotaxis (Ebrahimi et al., 2016). Such illustrates the considerable role of HSCs in hepatic fibrosis development, which has become an excellent target when developing antifibrotic agents useful for treating LF.
Hepatocytes are often the primary targets of viruses affecting the liver, alcohol, steatosis, and other hepatotoxic agents. Chronic forms of hepatic injury contribute to hepatocytes’ programmed cell death mainly via Tumour Necrosis Factor-α (TNF-α) associated apoptosis. During the cirrhotic stages, hepatocytes act as the chief source of TGF-β, enhancing fibrogenesis in the liver (Ebrahimi et al., 2016). Under normal conditions, the LSECs regulate the movement of fluids and various particles between the hepatocytes and blood in the sinusoids. However, during liver injury, the LSECs become defenestrated and capillarized, converting them into activators of HSCs (Ebrahimi et al., 2016). Consumption of alcohol and viral hepatitis results in the activation of the Kupffer cells. Once activated, Kupffer cells release cytokines and express chemokine receptors enhancing the progression of LF. Besides, the activated Kupffer cells stimulate the HSCs (Ebrahimi et al., 2016). As such, hepatocytes, Kupffer cells, and LSECs also contribute to fibrosis in the hepatic systems, especially after a hepatic injury. Therefore, antifibrotic therapies should target the various roles of cells in the hepatic system to effectively prevent hepatic fibrosis’s formation and progression.
2.3.4 Diagnosis of LF
The measurement of most of the serum markers is a typical laboratory routine. Such markers often rise in the event of hepatic inflammation, making them nonspecific for the diagnosis of LF. Albumin is among the proteins synthesized by the liver. As such, albumin levels significantly reduce in the case of hepatic disease. However, such a decline in serum albumin levels often occurs in the case of chronic diseases such as inflammatory conditions and kidney diseases associated with excessive loss of proteins (Lurie et al., 2015). Therefore, even if albumin is an excellent hallmark of ill health, it is not specific to liver disease. Most laboratories use most of these markers in combination (Ahmad et al., 2011; Liu et al., 2012). Conversely, even though the combined biomarkers are often in use in clinical practice, especially in excluding advanced cases of hepatic fibrosis and cirrhosis, they cannot distinguish the early from the intermediate stages of fibrosis (Castera & Pinzani, 2010). Some of the biomarkers utilized in the diagnosis of hepatic fibrosis include the AST/ALT ratio, APRI, fibrotest, and FIB-4. The release of transaminases (ALT and AST) into the serum mainly occurs following destruction of the hepatocytes. When combined, the ratio of the two transaminases offers better data rather than focusing on each of the serum enzyme levels. FIB-4 takes into consideration the age of the patient, the serum levels of the two transaminases, and the platelet count. However, studies indicate that APRI is among the most straightforward markers that can effectively help clinicians to develop diagnosis LF and cirrhosis with excellent precision (Lurie et al., 2015). Fibrotest is among the best prognostic factors for liver diseases (Poynard et al., 2011). As such, it shows the potential of liver disease progressing to fibrosis and cirrhosis.
Among the various imaging modalities, MRI is the most common imaging technique that evaluates the liver for cirrhosis and its associated complications. Unfortunately, it does not pick the early stages of fibrosis. Computed tomography also reveals some of the significant morphological changes such as liver changes and signs indicating the presence of portal hypertension (Lurie et al., 2015). A liver biopsy helps in evaluating for LF. However, it has various limitations, with the first being its invasiveness. As such, there has been a necessity for developing accurate methods that are not invasive to effectively diagnose LF (Ebrahimi et al., 2016). Also, the combined biomarkers and imaging tools are an easier way of diagnosing hepatic fibrosis.
Fibroscan is among the most common test for evaluating patients for LF. Among its primary advantages compared to liver biopsy includes noninvasiveness, cheap, and it does not cause pain. Besides, it takes approximately five minutes to perform, unlike liver biopsy, where the patient must remain at the hospital for at least six hours (Afdhal, 2012). The fibroscope mainly works by estimating the shear wave velocity in the hepatic tissue. During the technique, a wave passes through the hepatic tissue from a transducer located on an ultrasound probe. Subsequently, there is the measurement of the shear wave’s velocity that is then converted to liver stiffness measured in kilopascals (Afdhal, 2012).
Fibroscan is useful for any patient when the clinical need is to determine the stage of LF. Some of the factors that might limit the effectiveness of a Fibroscan in assessing patients for hepatic fibrosis include the presence of ascites, morbid obesity, and patients who possess excess amounts of fat on the chest wall. In such instances, the results might be unreliable (Afdhal, 2012). As such, reliability and reproducibility play a substantial role in influencing the validity of elastography results when using a Fibroscan (Afdhal, 2012).
Other noninvasive methods of evaluating the liver for fibrosis include radiological investigations. Magnetic resonance elastography has high accuracy for detecting hepatic fibrosis by measuring the stiffness of the hepatic tissue. However, it requires time since a patient has to undergo a magnetic resonance imaging scan. Another investigation is the acoustic resonance force impulse testing, even though it is under evaluation and is currently yet to acquire approval for use in Europe and America (Afdhal, 2012). Clinicians often prefer the noninvasive methods for evaluating patients who present with features suggestive of hepatic disease culminating into hepatic cirrhosis.
Once a clinician collects the hepatic tissue using a biopsy, there is a histological analysis to determine the stage of fibrosis. Among the widely used staging systems is the METAVIR score. As per the score, there are four stages of LF. Stage 0 means there is fibrosis of the hepatic tissue. Stage 1 is when there is portal fibrosis, which does not involve any part of the septa. The next stage involves the fibrosis of the portal system and just a few septa. Entire septal fibrosis is the hallmark of Stage 3, while Stage 4 mainly features cirrhosis of the hepatic system (Suk & Kim, 2015).
Figure 2.3: Liver histopathology using Metavir scoring system. Image A represents Stage 1 through to Stage 4 denoted by image D. Source:(Li et al., 2015).
2.3.5 Potential therapeutic agents of LF
Among the primary source of fibrotic tissue are the myofibroblasts that mainly form from the activated HSC and fibroblasts from the portal system (Huang & Ogawa, 2012). Myofibroblasts are one of the cells which play an influential role in various organs, especially when it comes to wound healing. As such, their loss often contributes to necrosis of the tissue and initiation of the inflammatory process (Puche et al., 2013). Therefore, the treatment strategies under development should not aim at eliminating the myofibroblasts. Instead, the therapeutic agents should aim at reducing the fibrogenic activation of the myofibroblasts and stimulating the fibrolytic enzymes. Most of the agents developed to block the fibrogenic activation and the secretion of the EC matrix by the myofibroblasts show excellent results when used in cultures and some of the rodent models when targeting hepatic fibrosis. However, the same agents reveal a wide range of adverse effects to human beings secondary to their lack of specificity to target the specific cells only (Schuppan & Kim, 2013).
Presently, three main strategies are under study when focusing on targeting the function of the already activated myofibroblasts. One of the strategies aims at focusing mainly on the relevant pathways that culminate in the deposition of the matrix following the up-regulation of the myofibroblasts such as the procollagen 1 pathway and blocking the receptors of the various chemokines released as a consequence of the fibrogenic activation (Schuppan & Kim, 2013). However, the existing therapeutic agents that can effectively block collagen synthesis have widespread adverse effects since they affect other body organs. Imatinib, which acts by inhibiting the activity of tyrosine kinase, affects the fibrogenic signaling but further studies are essential primarily when focusing on hepatic fibrosis (Schuppan & Kim, 2013). PDGF blocking antibodies show a potential effect in reducing the development of hepatic fibrosis (Ogawa et al., 2010). As such, it is presently not in use as a treatment agent for liver fibrosis. The second strategy was targeting the myofibroblasts that were on the activation mode to apply the refined form of siRNA techniques, especially the liposomal formulations that deliver products to the myofibroblasts (Love et al., 2010). The last strategy involved the utilization of ligands, which were specific to receptors found on the activated myofibroblasts whose primary role was to enhance the efficacy of the drugs reducing the adverse effects by making them more specific (Schuppan & Kim, 2013). An example of the use of this strategy was the successful use of interferon-gamma in inhibiting the advancement of LF in mice (Bansal et al., 2011). Some of such therapeutic agents would require delivery systems such as pegylation to raise their half- lives (Pasquetto et al., 2011). Further studies on humans ought to take place to measure the efficiency of such agents in the management of LF among humans.
In the pathophysiology of LF, chemokines play a meaningful role in enhancing oxidative stress and apoptosis of hepatocytes (Wasmuth et al., 2010). Subsequently, the myofibroblasts play a unique part in facilitating the phagocytosis process of the dead hepatocytes resulting in their activation through the NOX2 pathway (Jiang et al., 2010). In connection with this, explorations show that the use of an agent that has the capability of inhibiting the apoptosis of hepatocytes or the use of agents that act as inhibitors of cathepsin B improved biliary fibrosis when used in mice (Schuppan & Kim, 2013). Such data shows some of the potential effective therapies for LF in humans.
Drugs that aim at the biliary fibrogenic progenitors have also shown some positive antifibrotic impacts on the biliary systems of rodents and the advanced form of fibrosis, which is not affecting the biliary system. Some examples of such drugs include therapeutic agents, mainly targeting a particular integrin in the biliary system that serves as a receptor for tenascin-C and fibronectin (Schuppan & Kim, 2013). Vismodegib, which is among some of the drugs that inhibit the hedgehog pathway, shows a potentially positive effect in the biliary fibrogenesis and cancer development (Philips et al., 2011; Syn et al., 2012). Some explorations indicate that the use of such hedgehog pathway inhibitors reversed cases of HCC in mice (Philips et al., 2011). Such agents show potential effectiveness in treating liver fibrosis in humans.
Monocytes are the primary precursors of Mφ(s), fibrocytes, and DC, explicating similar features to the suppressor’s cells of the myeloid lineage (Schuppan & Kim, 2013). As such, monocytes play a noteworthy part in enhancing the course of inflammation and, consequently, fibrosis. At the crossing points of the two types of immunity, which include innate and adaptive, the monocytes play a principal purpose in augmenting the immune responses from the adaptive immunity by the use of the proinflammatory monocytes (Zimmermann et al., 2010). During the recruitment of monocytes to the site of inflammation, chemokines, together with their receptive sites, serve the role of chemoattraction, enhancing the process of fibrosis modulation (Saiman & Friedman, 2012; Wasmuth et al., 2010). Studies among rodents show that chemokine CXCL 9 and 10 prevent fibrogenesis once they activate their receptor (Karlmark et al., 2010; Sahin et al., 2012). Other explorations designate that the action of other chemokines such as CCL2 improves fibrosis and enhances the slow reversal of the hepatic tissue from the scarring tissue (Ambade et al., 2019). Such facts indicate that agents that can enhance the production of these chemokines can prevent the advancement of LF.
The formation of novel vessels in the liver often occurs with the stimulation and proliferation of LSEC, which links closely with the deposition of the matrix on the perisinusoidal regions. During the process, LSEC also takes part in the production of the EC matrix that ultimately stimulates the HSC while secreting factors that facilitate vasoconstriction of the vessels in the liver, augmenting the portal pressures commonly seen in patients who have hepatic cirrhosis (Schuppan & Kim, 2013).
On the other hand, myofibroblasts stimulate the LSEC through the secretion of angiogenic agents such as the VEGF (Thabut & Shah, 2010). In connection with this, explorations indicate that therapeutic drugs that aim at inhibiting the angiogenic process show some mitigation effect from the progression of the hepatic disease to LF in models. Antifibrotic effects are evident among agents that have an additional impact of affecting other cellular components and the immune system, such as the polykinase inhibitors, which include sorafenib (Thabut et al., 2011). Such lack of specificity explains some cases of deterioration that are common when there is a combination of the anti-VEGF antibody with integrin antagonist (Schuppan & Kim, 2013; Stockmann et al., 2010). The reason behind such worsening is because the former agent mainly inhibits angiogenesis while the later inhibits both vessel and LSEC proliferation (Schuppan & Kim, 2013). As such, the use of antiangiogenic agents for patients who have hepatic fibrosis is variable.
An injury to the cholangiocytes often culminates into the activation of the inflammation signaling pathways occasioning in the conscription of cells from the bloodstream. The injury also contributes to the stimulation of the fibroblasts in the portal system, which makes them release elements that facilitate the activation of the progenitor cells in the biliary system. When the injury is iatrogenic, especially during the ligation of the bile duct, the cells of the biliary system tend to express the hedgehog ligands and receptors (Wang et al., 2016). The hedgehog ligands also come from the activated myofibroblasts in the biliary system. Studies indicate that agents that target to block the signaling of the hedgehog pathway such as GDC-0449, could play an essential function in blocking the myofibroblasts even in the hepatic system from releasing the matrix, which would lead into the formation of fibrotic tissue (Wang et al., 2016). The same agent shows a positive effect in enhancing the reversal of hepatocellular carcinoma in animal models. Following the ligation of the biliary duct, some of the cholangiocytes indicate an excessive expression of integrin αvβ6, which proposes that the inhibition of the integrin would play a unique function in preventing the progression and formation of the fibrotic tissue in the hepatic system. Studies indicate that a single dose of drugs that inhibit the activity of integrin αvβ6 shows positive antifibrotic effects in mice (Wang et al., 2016). In line with this, it is fundamental to develop therapeutic agents that focus on inhibiting the action of myofibroblasts and Integrin αvβ6. This calls for further studies to decide the effectiveness of such drugs in the management of patients diagnosed with hepatic fibrosis since most of the studies involved animal models rather than humans.
The best effective therapy for hepatic fibrosis would be the eradication or the treatment of primary liver disease, which will ultimately result in the deposition of the extracellular matrix. However, this is not always feasible since most of the time, patients are often asymptomatic, often presenting to the hospital when there is already a progression of the chronic liver disease into fibrosis.
One of the major drugs commonly utilized in the management of most of chronic liver diseases include steroids (Altamirano-Barrera et al., 2017). An exploration carried out by Zhao and colleagues aimed at determining the efficacy of utilizing steroids in the management of patients who had the acute form of liver disease. The study revealed that the use of steroids as part of the regimens for patients who had acute liver failure raised their survival rates since it had an effect on slowing the overall advancement of the disease and occasioned in the capacity of the liver to regenerate (Zhao et al., 2016). Another exploration confirmed similar results by indicating a histological improvement among patients suffering from the autoimmune form of hepatitis who were on steroids. The study also publicized a decline in the rate of fibrosis among those patients treated with steroids during at least four months. The histological activity score among the participants of the study also improved. Another finding in the study was the fact that when focusing on hepatic fibrosis, roughly eighty percent of the patients who received the corticosteroids showed the lack or reversal of hepatic fibrosis (Altamirano- Barrera et al., 2017). When used among patients with autoimmune hepatitis, steroids mainly offer the advantage of limiting further injury to the hepatic tissue, negatively affects the signaling pathways which enhance the formation of fibrosis, and augment the breakdown of the ECM, which are vital in the formation of LF (Montano-Loza et al., 2016).
Another finding in one of the studies was the effect of caffeine on hepatic fibrosis. Caffeine is one of the widely known purine alkaloids that act via the adenosine receptors, where it serves as an opponent of the type A1 and A2A receptors. Mostly, the overall effect of caffeine takes place when its therapeutic concentration is between ten and a hundred micromolar (Altamirano-Barrera et al., 2017). A study carried out by Modi and colleagues aimed at unveiling the relationship that exists between the intake of caffeine and hepatic fibrosis. During the exploration, patients going through a liver biopsy would take part in filling in questionnaires that mainly contained questions related to caffeine over six months (Modi et al., 2010). The outcomes of the investigation revealed that regular coffee intake of above at least two cups daily contributed to a less severe form of LF (Modi et al., 2010). Such studies illustrate the health benefits of regular use of coffee, unlike most of the previous explorations, which would indicate the widespread adverse health effects of coffee consumption. Altamirano-Barrera et al. (2017) postulate that the regular intake of caffeine offers some health benefits to patients suffering from hepatic fibrosis by blocking the activity of adenosine, which is among the most potent endogenous product that controls the inflammatory pathways and tissue repair. HSC often expresses the caffeine receptors type 2A, whose stimulation culminates in the production of collagen. Caffeine also shows positive effects among patients suffering from hepatic steatosis (Altamirano- Barrera et al., 2017).
In line with this, one of the investigations reveals that the severity of steatosis was higher among people who consumed lower amounts of coffee (Altamirano-Barrera et al., 2017). The same findings appear among animal models with a fatty liver where the intake of coffee often contributes to raises resistance of insulin at the hepatic level and decreases the overall production of inflammatory cytokines (Gutiérrez-Grobe et al., 2012).
UDCA is another product that has shown antifibrotic effects for quite a long time. Over the past years, UDCA has been useful in the management of patients who present with features of primary biliary cholangitis, which enables the delay of the process of liver transplantation. Among patients who receive the therapeutic agent, there was a decline in the progression of hepatic fibrosis, revealing that UDCA affects LF (Altamirano-Barrera et al., 2017). The action of hindering the progression of LF among patients who are suffering from primary biliary cholangitis was more pronounced when the healthcare providers combined UDCA with budesonide (Cheng & Wong, 2017). Besides, UDCA, in combination with candesartan, which acts by blocking the angiotensin receptor, revealed better improvement of the hepatic fibrosis as compared to when patients took UDCA as a single drug for six months (Kim et al., 2012). In connection with this, investigators should take part in carrying out more explorations that would ascertain the effectiveness of such therapeutic agents in humans while assessing their adverse effects.
Obeticholic acid, which is an analog of the chenodeoxycholic acid working by stimulating the farnesoid X receptor, shows some positive antifibrotic and anti-inflammatory effects (Mudaliar et al., 2013). As such, it is under scientific trial for the management of LF, as shown in Tables 4.1 and 4.2 below. Research shows that obeticholic acid plays a significant part in improving the liver biochemistries among patients with LF, indicating that it enhances the anti-inflammatory activity and hepatic injury, which leads to a drop in the serum levels of transaminases (Nevens et al., 2016). The farnesoid X receptor is among one of the widely researched nuclear transcription factors whose stimulation mainly occurs following its conjugation with the bile acids (Altamirano-Barrera et al., 2017). Chenodeoxycholic acid has also shown some antifibrotic effects in animals even though its effect is lower compared to that of the obeticholic acid. One of the studies revealed that animals that received approximately one to ten milligrams per kilogram of chenodeoxycholic acid for the three months showed a decline in the expression of collagen type one, which would ultimately result in a decline in hepatic fibrosis (Altamirano-Barrera et al., 2017).
Another group of therapeutic agents under research are those that target endocannabinoids, which are mainly composed of lipid molecules acting via the Cannabinoid Receptor Type 1 (CB1).
An upsurge in the action of the receptor plays a noteworthy part in the advancement of visceral obesity, which often brings about the development of hepatic fibrosis, DM, and NAFLD (Dibba et al., 2018; Jourdan et al., 2013; Silvestri & Di Marzo, 2013). Research shows that the stimulation of the type 1 receptor augments the manifestation of profibrotic genes, which ultimately enhances the establishment of LF (Altamirano-Barrera et al., 2017). As such, agents that block the receptor would culminate in preventing the formation of LF. Unfortunately, the use of such agents that block the endocannabinoid receptor type 2 often results in neuropsychiatric adverse effects.
Another critical element that plays a significant function in stimulating the formation of LF is the inducible form of nitric oxide synthase. Studies show that an upsurge in the action of the enzyme often augments the advancement of viral hepatitis, culminating into hepatic fibrosis (Navarro et al., 2015). A study carried out by Cinar and colleagues developed a hybrid therapeutic agent that could inhibit the function of both the CB1 receptor and the inducible form of the enzyme in mice, making a potentially effective therapy for enhancing the management of patients with hepatic fibrosis (Cinar et al., 2016). Such a hybrid drug would be quite valuable in reducing the degree of advancement of hepatic fibrosis since it works by targeting two diverse sites that enhance the formation of LF.
Antioxidants such as Vitamin E also play a substantial function in managing patients with LF even though their use remains controversial. A study carried out by Sanyal and colleagues aimed at determining the best therapeutic agents for patients who were suffering from NASH. In line with this, the exploration involved 247 non-diabetic participants suffering from NAFLD who received pioglitazone, vitamin E or a control agent for two consecutive years, aiming at determining whether there would be any histological improvements of NASH (Sanyal et al., 2010). The findings of the investigation indicated that individuals given vitamin E had improved outcomes of NAFLD (Sanyal et al., 2010).
Stellate cells in the hepatic system are the primary cells whose purpose is to produce collagen, and their ultimate stimulation is among the critical steps in the formation of hepatic fibrosis. As such, therapeutic agents that target the HSC would be potent drugs for managing patients who develop hepatic fibrosis (Cheng & Wong, 2017). According to Huang et al. (2017), a reversal of hepatic fibrosis can effectively occur via the use of agents that can block the stimulation of HSC, agents that can augment the immune clearance of the stellate cells, and agents that can enhance the apoptosis of the HSC. Some of the agents that have shown a possible effect in blocking the instigation of the stellate cells in the hepatic system include PPAR- γ agonists such as pioglitazone’s (Cheng & Wong, 2017).
In the pathophysiology of LF, chemokines play a meaningful role in enhancing oxidative stress and apoptosis of hepatocytes (Wasmuth et al., 2010). Subsequently, the myofibroblasts play a unique part in facilitating the phagocytosis process of the dead hepatocytes resulting in their activation through the NOX2 pathway (Jiang et al., 2010). In connection with this, explorations show that the use of an agent that has the capability of inhibiting the apoptosis of hepatocytes or the use of agents that act as inhibitors of cathepsin B improved biliary fibrosis when used in mice (Schuppan & Kim, 2013). Such data shows some of the potential effective therapies for LF in humans.
Drugs that aim at the biliary fibrogenic progenitors have also shown some positive antifibrotic impacts on the biliary systems of rodents and the advanced form of fibrosis, which is not affecting the biliary system. Some examples of such drugs include therapeutic agents, mainly targeting a particular integrin in the biliary system that serves as a receptor for tenascin-C and fibronectin (Schuppan & Kim, 2013). Vismodegib, which is among some of the drugs that inhibit the hedgehog pathway, shows a potentially positive effect in the biliary fibrogenesis and cancer development (Philips et al., 2011; Syn et al., 2012). Some explorations indicate that the use of such hedgehog pathway inhibitors reversed cases of HCC in mice (Philips et al., 2011). Such agents show potential effectiveness in treating liver fibrosis in humans.
Monocytes are the primary precursors of Mφ(s), fibrocytes, and DC, explicating similar features to the suppressor’s cells of the myeloid lineage (Schuppan & Kim, 2013). As such, monocytes play a noteworthy part in enhancing the course of inflammation and, consequently, fibrosis. At the crossing points of the two types of immunity, which include innate and adaptive, the monocytes play a principal purpose in augmenting the immune responses from the adaptive immunity by the use of the proinflammatory monocytes (Zimmermann et al., 2010). During the recruitment of monocytes to the site of inflammation, chemokines, together with their receptive sites, serve the role of chemoattraction, enhancing the process of fibrosis modulation (Saiman & Friedman, 2012; Wasmuth et al., 2010). Studies among rodents show that chemokine CXCL 9 and 10 prevent fibrogenesis once they activate their receptor (Karlmark et al., 2010; Sahin et al., 2012). Other explorations designate that the action of other chemokines such as CCL2 improves fibrosis and enhances the slow reversal of the hepatic tissue from the scarring tissue (Ambade et al., 2019). Such facts indicate that agents that can enhance the production of these chemokines can prevent the advancement of LF.
The formation of novel vessels in the liver often occurs with the stimulation and proliferation of LSEC, which links closely with the deposition of the matrix on the perisinusoidal regions. During the process, LSEC also takes part in the production of the EC matrix that ultimately stimulates the HSC while secreting factors that facilitate vasoconstriction of the vessels in the liver, augmenting the portal pressures commonly seen in patients who have hepatic cirrhosis (Schuppan & Kim, 2013).
2.4: Conclusion
The literature review under this section mainly focuses on the chief knowledge of hepatic fibrosis, focusing on the existing research. The section offers some critical background data concerning hepatic fibrosis, including the epidemiology, diagnosis, and pathophysiology of the condition, which is particularly essential to understand how the effective therapeutic agents work in preventing the advancement of LF and enhancing the reversal of the fibrosis formation. The search strategy was quite effective in ensuring that there was an acquisition of credible journal articles that focused on the topic of this dissertation. This chapter plays an imperative role in unveiling the existing research gap, which will play a pivotal function in guiding the research, analysis, and development of the following chapters. Focusing on the five databases made the work easier to retrieve credible and authentic information that could be part of this literature review section.
Chapter 3: Methodology
3.1 Introduction
This segment mainly aims at illustrating how the exploration will take place. It reveals the essence of carrying out the study compared to any of the prevailing investigations. In this section, there will be an inclusion of the research design and the method utilized in data collection. It will also include some of the significant limitations encountered when carrying out the study. An ethics section will also be present to confirm if there was any ethical approval when carrying out the exploration. A conclusion encompassing a summary of the main ideas presented in the methodology sections will also be present at the end of this chapter. This section aims at indicating the various aspects of the study, including how the investigator takes part in the study, making it easier for the reviewer to quickly comprehend the sequence of the exposition, including the design and data collection modalities.
3.2. Research Design
The design of the exploration is a crucial part when working on a dissertation in science and other disciplines. Following the identification of the topic of the dissertation and developing the research questions, the investigators must select the best design to utilize during the study to make sure that the entire process becomes a success. Presently, there is a superfluity of literature that offers some guidance on how one should select the research design of an exploration. However, such knowledge at times brings about some confusion among scholars in various fields since the data does not offer similar guidelines lacking the essential clarity (Abutabenjeh & Jaradat, 2018). This is qualitative systematic review research that involved the analysis of numerous primary and secondary sources related to the topic “Development of effective therapies against liver fibrosis”.
3.3 Data Collection Method
The main data collection technique involved analyzing currently existing statistics from previous studies. The analysis involved one hundred and twenty journal articles derived from the five major databases, including Science Direct, PubMed, Web of Science, Cochrane Library, and the BMC. A systematic qualitative review mainly entails finding existing explorations on a particular topic and systematically reviewing the evidence provided to enable one to conclude the topic at hand (Seers, 2015). Debate exists as to whether the search must always be exhaustive (Campbell et al., 2011; Toye et al., 2013)). When working on this dissertation, there was a selection of the most relevant articles, even though it was not fully exhaustive. The selection of the journal articles from the various databases ensured that the data collected for analysis would be authentic, valid, and accurate. The analysis used articles published within the last ten years to offer some guidance on some of the developments that have taken place in the process of developing therapies targeting LF in line with the topic.
3.4 Limitations
Among the major limitations when using the systematic review process of acquiring data is the challenges faced when searching and screening the various sources to determine the specific articles which will be useful when carrying out the study (Mallett et al., 2012). For example, when carrying out this dissertation, there was a wide range of journal articles containing essential data for this research. Still, the process could only accommodate just an ample number. Such a process could also have been a contributor of bias. Systematic reviews often require a vast range of access to numerous databases, some of which might require a subscription for one to acquire some of the articles. Such an activity can be quite expensive (Mallett et al., 2012). However, to overcome this limitation, most of the journal articles utilized when carrying out the exploration were mainly accessible freely on the various online search engines. Such a strategy ensures that any reader going through the dissertation can easily access the primary source of the data in case they need to delve into further research. Even though there were well-defined inclusion and exclusion criteria, it was quite challenging to note that some of the journal articles which seemed to contain a wide range of information fell under the exclusion group. For example, there were some articles published more than ten years ago that contain relevant data on the topic concerning the management of liver fibrosis. Such articles would be essential in providing some basis of the topic to ensure that a comparison of such data with the current articles reveals if there has been any progress in mounting effective treatment options for LF. However, the criteria excluded such articles as part of the sources for this study.
3.5 Ethics
Most of the journal articles utilized when working on this dissertation revealed that the investigators had acquired the necessary ethical approval from the relevant Ethical Committees. The articles maintained the anonymity of the patients who participated in the studies while ensuring that they acquired informed consent from all their participants. Other articles did not include any ethical approval confirmation significantly the systemic review articles. When carrying out this systemic review form of study, there was no need for ethical approval.
3.6 Conclusion
The methodology of any exploration plays an influential role in determining its relevance and how it achieves its primary objectives and aims. As such, there was a sacrifice of much energy and time to develop a superb methodology. Out of the one hundred and twenty articles derived from the five databases, forty of them were literature reviews, thirty-seven of them were systematic reviews, twenty-three of them were empirical studies, and the remaining ones were meta-analyses. It is quite evident that the LR is satisfactory and aligns with the methodology with the primary purpose of achieving the research aims and objectives.
Chapter 4: Data Analysis
4.1 Introduction
This segment mainly presents the main findings developed after analyzing the wide range of journal articles from the five reputable databases. Having generated an in-depth literature review on chapter one of this dissertation and an excellent methodology in chapter two, this section aims at bridging together all the data acquired from various articles. The section includes several tables and other graphical presentations that aim at representing the main findings of the study.
4.2 Analysis Strategy
The exploration involved an analysis of one hundred and twenty journal articles acquiring the data presented below. The analysis mainly contains information derived from sources published in the past ten years. In line with this, it makes it easy for the analysis to illustrate some of the developments that occurred in the area of searching for effective therapies for managing patients who develop hepatic fibrosis. The utilization of primary and secondary articles as the source of data might seem to be cumbersome at times since one has to utilize most of their time and energy in understanding the primary ideas and themes of every article. Another pitfall of using this strategy is that in case some investigators did not thoroughly analyze their works, there is a likelihood of including such data which can result in bias.
4.3: Presentation and Discussion
4.3.1 Eradicating underlying disease
When treating patients with LF, the initial step is to focus on managing the underlying liver disease, as shown below in Figure 4.1. Most patients with chronic cases of viral hepatitis often develop hepatic fibrosis. Currently, there are numerous effective antiviral drugs that enhance the regression of LF and cirrhosis, especially following a sustained suppression of the viral load or after clearing the virus from the bloodstream (Cheng & Wong, 2017). The study carried out by Marcellin et al. (2013) involved one year during which there were randomized clinical trials using TDF and adefovir dipivoxil. Subsequently, the participants would take part in the exploration for over seven years using the TDF treatment. Among the participants, some of them had hepatitis B disease while others were negative and served as the controls. A repeat liver biopsy was to take place after five years to assess whether there was any improvement. The exploration revealed that among patients who had Hepatitis B infection, especially the chronic form, managing with TDF for at least five years was effective and innocuous. Such a period of suppressing the viral load also led to the reversal of LF and cirrhosis (Marcellin et al., 2013). Another exploration carried out by Schiff et al. (2010) involved patients who had advanced stages of LF and cirrhosis and had to receive entecavir for a minimum of three years. The Ishak fibrosis score enabled the investigators to determine whether there was any improvement in LF after the treatment. The results indicated that patients who had CHB showed significant improvement in their LF status following a long-term period of taking entecavir. The drug also enhanced the reversal of LF and cirrhosis to a great extent (Schiff et al., 2011). Some Chinese and Korean cohorts took part in exploring the purpose of metabolic factors in enhancing the reversal of LF. Metabolic syndrome consists of a constellation of medical conditions such as DM, HTN, central obesity, and dyslipidemia (Yoon et al., 2013). The exploration aimed at unveiling the relation between metabolic syndrome and the development of LF among patients who already had CHB. The outcomes of the research revealed that metabolic syndrome would independently cause the development of LF and progression to cirrhosis among patients suffering from CHB (Yoon et al., 2013). Another exploration carried out by Wong et al. (2014) revealed the same findings showing that the presence of metabolic syndrome would increase the risk a patient to develop LF, especially if they had CHB, no matter the viral load in the bloodstream or the viral action (Wong et al., 2014). According to Cheng et al. (2016), it is imperative to control metabolic syndrome among patients diagnosed with CHB even if they have sufficient viral suppression since it will not only enhance the reversal of LF but will also minimize mortalities of such patients, most of which emanated from cardiovascular complications (Cheng et al., 2016).
Figure 4.1: Major underlying causes of LF. Sources: (Wiegand & Berg, 2013).
As shown in Figure 4.1 above, another major cause of LF is CHC infection. Explorations indicate that the use of conventional interferon played a noteworthy part in enhancing the regression of LF among patients who had chronic hepatitis C infection (Cheng & Wong, 2017). Another exploration revealed a remarkable reversal of hepatic fibrosis occurring in more than eighty percent of the participants, enhancing the recovery of the hepatic architecture to almost normal or near-normal (Cheng & Wong, 2017). Currently, there has been widespread utilization of the DAA agents, such as sofosbuvir, as part of management for hepatitis C viral chronic infection where they achieve an excellent virological response (Jakobsen et al., 2017). Most of the studies evaluating the reversal of hepatic fibrosis among patients with CHC taking DAA agents do so by the utilization of non-invasive strategies such as the use of transient elastography, which estimates the liver stiffness. One of the explorations carried out by Knop and colleagues involving fifty-four cirrhotic patients who were on DAA agents revealed a slight reduction in the stiffness of the hepatic tissue since the baseline measurements compared to the post-treatment measurements (Knop et al., 2016). Such findings made the investigators conclude that the decline in the liver stiffness evaluation was secondary to decreased necroinflammation and a slight reversal of cirrhosis (Knop et al., 2016).
Another contributor of hepatic fibrosis includes metabolic factors. As such, controlling the metabolic factors is imperative when managing patients with NAFLD, just like eradicating the hepatitis virus.
Studies indicate that working on reducing the bodyweight of at least ten percent or more through lifestyle modifications such as taking a healthy balanced diet and taking part in regular physical activities enhances the regression of hepatic fibrosis, especially in the mild and moderate cases (Vilar-Gomez et al., 2015; Wong et al., 2013). Thiazolidinediones such as pioglitazone, which act as agonists of the PPAR-γ and commonly used for the management of diabetes mellitus, reveals some positive impacts in enhancing the reduction of LF in some meta-analyses studies (Boettcher et al., 2012; Chang et al., 2013; He et al., 2016; Mahady et al., 2011). Nevertheless, the outcomes of some of the recent studies did not confirm the findings (Singh et al., 2015). Another exploration is done on pentoxiphylline, a derivative of xanthines, and obeticholic acid revealed an excellent decrease of hepatic fibrosis, especially among people who had NAFLD (Neuschwander-Tetri et al., 2015).
Among patients with NASH, fibrosis is among one of the most widely applied prognostic factors. As such, there have been various studies done, aiming at developing antifibrotic agents (Cheng & Wong, 2017). Wong and colleagues explored patients suffering from NAFLD, where there was an analysis of Simtuzumab, an agent that aims at the LOXL2, which was in Phase 2 trials as an agent for treating NAFLD among patients who had extensive LF and cirrhosis (Wong et al., 2016). LOXL2 is an enzyme whose primary role is to enhance the linkage of ECM proteins once deposition occurs, facilitating the process of fibrosis (Moon et al., 2014). Unfortunately, the company which was taking part in the drug trial announced the termination of the trials after the drug failed to show any efficacy against both NASH and PSC during the second phase of the trials (Cheng & Wong, 2017). A contemporary study carried out by Loomba and colleagues indicates that the use of ASK1 inhibitors such as selonsertib plays a momentous part in enhancing the regression of fibrosis and inflammatory reactions in the liver using animal models (Loomba et al., 2018). The drug reduced the overall hepatic stiffness, and there was a reduction in the levels of the serum biomarkers of apoptosis. As shown in Table 4.1, there are numerous clinical experiments ongoing to define the efficiency of the drug in managing patients who have hepatic fibrosis, mainly focusing on those patients whose primary cause of LF and advanced cirrhosis is NASH.
As shown in Table 4.1 below, cenicriviroc is another therapeutic agent that is presently under trial for dealing with LF. It serves as an antagonist of CCR5 receptor. As such, cenicriviroc negatively impacts the inflammatory process taking place in patients with NAFLD. When using animals as the sample models, the drug has displayed positive anti-inflammatory and antifibrotic impacts (Berres et al., 2010; Lefebvre et al., 2016). Some other drug developments which are under trials for the treatment of LF, as shown in Table 4.1 and Table 4.2., are the galectin inhibitors such as GR-MD-02. The therapeutic agents act by binding to the galactose residues in the glycoprotein, which serves to enhance a reduction of hepatic fibrosis among animal models (Traber et al., 2013).
Table 4.1: Some of the active trials on anti-fibrotic agents. Source: (Cheng & Wong, 2017).
According to Bansal et al. (2016), the widespread advancements in researchers developing a proper understanding of the molecular basis resulting in hepatic fibrosis offered promising molecular targets for the growth of antifibrotic agents. Nonetheless, one of the challenges to such potential developments has been the slow progression of hepatic fibrosis among humans, and the non-invasive techniques utilized to evaluate the progression of hepatic disease often lack the required sensitivity. Besides, studies show that most patients who have hepatic fibrosis are often asymptomatic even when the condition is in the advanced stages when the survival rate is low (Bansal et al., 2016). As such, by the time most patients are presenting with any signs or symptoms indicating that there is a liver disease, it is too late to utilize such drugs. The development of the drugs that have antifibrotic effects would thus require the investigators to take into account both long and short-term therapies. The drugs can either be in the form of antibodies or small molecule inhibitors (Bansal et al., 2016). Antibodies have the disadvantage of having a lower bioavailability as such, not thoroughly recommended by most researchers (Ovacik & Lin, 2018). When developing therapeutic agents, it is essential to target a specific pathway in the molecular pathway of pathogenesis of hepatic fibrosis to ensure that the drugs are effective and also this would avoid any drug-to-drug interactions that might result in detrimental effects to the patient receiving the medications.
Among the final steps of LF is the secretion of the extracellular matrix, which mainly consists of excessive amounts of collagen types 1 and III, which is a result of injury to the liver (Tacke & Trautwein, 2015).
Activated HSCs show a potential effect in enhancing the rebuilding of the architecture of the liver following a period of hepatic fibrosis since they enhance the secretion of tissue metalloproteinases (Puche et al., 2013). In addition to modifying the HSC in a manner that they can act as effective therapies for LF, some drugs directly inhibit collagen synthesis, such as halofuginone, which enhances the complete resolution of hepatic fibrosis (Pines, 2014). However, some of such drugs tend to be nonspecific inhibiting collagen formation not only in the hepatic tissue but also in other organs such as the skin, which can have detrimental effects.
Table 4:2: Some of the existing clinical trials for the management of LF. Source:(Poilil Surendran et al., 2017).
Table 4.3: Some of the nanoparticles for the management of LF. Source: (Poilil Surendran et al., 2017).
4.3.2 Antifibrotic agents
4.3.2.1 Inhibitors of immune and inflammatory reactions
Among the primary potential therapeutic agents that could offer effective treatment for patients suffering from hepatic fibrosis are the antifibrotic agents. Most of these agents depict their action of enhancing the regression of hepatic fibrosis by mainly targeting the stellate cells in the liver in diverse ways. The first group agents mainly aim at reducing the signaling pathways of inflammation and the immune responses affecting the whole cascade, which would otherwise enhance the formation of LF. Among the commonly used agents under this group are glucocorticoids such as the steroids, whose primary function is to target the immune mediators, ultimately influencing the inflammatory pathways. Another agent that has the potential of affecting the regression of hepatic fibrosis is the consumption of caffeine. However, even though some studies do propose that regular coffee consumption may result in enhancing the reduction in the formation of hepatic fibrosis, other explorations claim against such ideologies. Caffeine acts via the adenosine type A2A receptor via which it affects the role of myofibroblasts in the process of LF formation by affecting the production of matrix proteins (Nathwani et al., 2019). Curcumin is another agent under this group of agents that mainly acts through the cannabinoid receptors, whereby downregulating the action of this receptor, the drug prevents the progression of hepatic fibrosis since the receptor serves the function of a profibrotic mediator. Silymarin is another therapeutic agent under trial that could fit into this group of drugs. The agent mainly targets the oxidative stress pathways, which it inhibits, resulting in the prevention of the activation of the stellate cells in the hepatic system. Ursodeoxycholic acid also falls among this group of potential therapeutic agents for managing hepatic fibrosis, and it mainly works by reduction of the cytotoxic effects of the bile acids, which would otherwise result in apoptosis of the liver cells (Nathwani et al., 2019).
4.3.2.2 Blockers of HSC stimulation
Another group of potential therapeutic agents for treating patients who develop hepatic fibrosis are the agents that block the process, which would otherwise lead to the activation of the stellate cells in the hepatic system. One such agent is vitamin E, which mainly works via preventing the pathways that participate in the enhancement of the oxidative stress, ultimately affecting the progression of LF. Another group of drugs that fall under this category is the thiazolidinediones, such as rosiglitazone. The drugs mainly work via the inhibition of the PPAR, which ultimately blocks the pathways that result in inflammation and formation of hepatic fibrosis (Nathwani et al., 2019).
The effect on fibrosis is mostly higher among patients who have LF that is mainly secondary NASH. Another group of drugs that show the potential of blocking the activation of the stellate cells in the hepatic system include the angiotensin-converting enzyme (ACE) inhibitors. Several studies have explored the antifibrotic function of ACE inhibitors (Nathwani et al., 2019). Most patients developing the chronic form of hepatic disease often have high levels of angiotensinogen and angiotensin-1 in the liver cells, especially in the hepatocytes. Research shows that the receptors for angiotensin II undergo hyperstimulation when one develops chronic hepatic injury, which ultimately causes the stimulation of the HSC. During the chronic hepatic injury, there is also the stimulation of the pituitary-adrenal axis, which enhances the pathway coding for the oxidative stress and inflammatory reactions, culminating in the establishment of hepatic fibrosis (Nathwani et al., 2019). As such, the use of ACE inhibitors is an excellent potential treatment option for patients with LF.
Another group of drugs that fall under this category is the tyrosine kinase inhibitors such as Imatinib. Research shows that even though Imatinib initially targeted treating patients who developed chronic myeloid leukemia and other gut stromal tumors, it is amongst the most compelling therapeutic agents for the management of hepatic fibrosis given that it has an effect of blocking the stimulation of the PDGF which plays a special function in the formation of LF (Qu et al., 2015). Imatinib was not only shown to affect the prevention of fibrosis formation but also augmented the reversal of fibrosis in animal models who have systemic sclerosis. The same effects were present in the pulmonary and hepatic systems (Qu et al., 2015). All the antifibrotic effects on the hepatic system following the administration of Imatinib primarily occur due to its capability of suppressing the stimulation of the stellate cells in the hepatic system. The action of blocking the activation of HSC improved when the animal models received both Imatinib and an ACE inhibitor, such as perindopril, which by itself plays a substantial function of targeting and impeding the expression of the TGF-beta 1 (Qu et al., 2015). As such, the two agents tend to reveal synergistic effects, which is more appealing than when the animal model receives each of the therapeutic agents separately. One of the studies also indicates that Imatinib has a remarkable effect in reducing the overall expression of some of the most imperative markers of fibrosis (Westra et al., 2014). However, it is indispensable to note that unlike sorafenib, which works on both decreasing the formation of hepatic fibrosis and hindering the progression of the fibrosis in the subsequent months and years, Imatinib only facilitates the former. Among the significant reasons for this is the fact that models that received Imatinib revealed approximately half a decline in the activity of matrix metalloproteinase in the stellate cells of the liver while leaving quite a considerable number of HSC still in active mode (Qu et al., 2015). Another agent that falls under this category of therapeutic agents that play a unique part in blocking the stimulation of stellate cells in the liver is recombinant interleukin 22. Studies indicate that interleukin 22 plays a significant function in the prevention of the process that can lead to liver injury (Kong et al., 2013). It mainly prevents damage to the hepatocytes while promoting the stimulation of pathways that result in the proliferation of the hepatocytes. Stimulation of the pathways often culminates into the upregulation of various anti-apoptotic genes, such as the B-cell lymphoma 2 genes. The hepatoprotective effect of the interleukin 22 is present in another study where the mice that received the genetically modified interleukin 22 revealed an extraordinary level of resistance to the liver injury induced using Concanavalin A due to the expression of the interleukin 22 in the animal model (Kong et al., 2013). Conversely, the mice that never received the genetically modified form of interleukin 22 showed extensive liver injury. The administration of the interleukin 22 attenuated the ethanol-induced lipogenesis, which otherwise has resulted in hepatic steatosis, causing the formation of hepatic fibrosis. The animal models that received the genetically modified form of interleukin 22 revealed a down surge in the serum quantities of transaminases, principally ALT and AST, which was secondary to the interleukin preventing the injury to the hepatocytes. Mice that received the recombinant form of interleukin 22 did not express any form of liver injury that would often take place on the administration of acute ethanol challenge. Therefore, there would be a blockage of the pathway resulting in the formation of hepatic fibrosis in such models. Administration of an antibody that inhibits the activity of the interleukin 22 showed a decline in the overall proliferation of the hepatocytes, especially if it happened before carrying out a partial hepatectomy (Kong et al., 2013). As such, the recombinant form of interleukin 22 would be a prospective therapeutic agent for handling patients diagnosed with LF, especially the one which develops secondary to alcohol consumption.
 Figure 4.2: Some nanoparticles used for treating LF. (Poilil Surendranet al., 2017).
Figure 4.2: Some nanoparticles used for treating LF. (Poilil Surendranet al., 2017).
Figure 4.3: Modifications of nanoparticles to target specific hepatic cells. (Poilil Surendran et al., 2017).
The ROCK (Rho-Kinase) inhibitor is another agent that can effectively fall under the category of therapeutic agents that have the potential of enhancing the treatment of individuals diagnosed with LF by impending the stimulation of the HSC. Prior studies indicate that ROCK inhibitors have the capability of functioning towards the blockage of the stimulation of the stellate cells in the hepatic system, which eventually enhances the attenuation of hepatic fibrosis over time (Okimoto et al., 2019). However, the systemic effects that often occur as a consequence of the administration of the ROCK inhibitor outweigh its benefits. As such, one of the explorations carried out by Okimoto and colleagues aimed at developing a therapeutic agent that could possess the potency and capabilities of the ROCK inhibitors but would not have similar widespread adverse effects as the original ROCK inhibitors (Okimoto et al., 2019).
In connection with this, the developed agent would selectively block the stimulation of the hepatic cells in the liver while causing minimal side effects. The investigators developed an agent that contained the ROCK inhibitor, which would couple with vitamin A using liposomal bonds.
The use of nanoparticles incorporates liposomes, as shown in Figures 4.1 and 4.2 above. Mice that received the vitamin A coupled agent would ultimately express a momentous enhancement in their hepatic fibrosis, and they did not show any of the severe systemic effects seen on administering the ROCK inhibitors alone (Okimoto et al., 2019). For example, the renal function tests of the mice that received the modified form of the ROCK inhibitors were normal as opposed to the deranged kidney function tests among the animal models that received the non-modified version of the ROCK inhibitors. Besides, the animal models that received the modified form of the ROCK inhibitors also revealed a tremendous decline in their serum ALT and AST levels indicating that the vitamin A coupled agent would prevent the injury to the hepatocytes, which would otherwise augment the formation of hepatic fibrosis (Okimoto et al., 2019). As such, modified ROCK inhibitors have the potential as therapeutic agents for treating patients who develop hepatic fibrosis.
Oleoylethanolamide (OEA) is another therapeutic agent that has a significant potential to become one of the effective therapies for the management of hepatic fibrosis. In an exploration carried out by Chen and colleagues, the aim was to determine whether the capability of the OEA and the mechanisms it uses to attenuate hepatic fibrosis focusing on mice that had LF induced using thioacetamide (Chen et al., 2015). The animal models would then receive a daily dose of the therapeutic agent via the intraperitoneal route. The outcomes of the research confirmed that OEA mainly enhanced the prevention of progression of hepatic fibrosis by targeting to effectively block the stimulation of the stellate cells of the liver. Molecular analysis of the mice would further unveil the fact that OEA would also act by blocking the stimulation of the alpha-smooth muscles together with the collagen matrix (Chen et al., 2015). The muscles are an essential part in the process of the activated myofibroblasts laying the collagen and other matrix proteins. The drug would also inhibit the pathways involved in the process of inflammation and remodeling of the ECM. The exploration also indicates that the agent also works by blocking the stimulation of the transforming growth factor in the animal models. All these mechanisms are the ones that explain how OEA enhanced the amelioration of hepatic fibrosis. OEA has also shown positive effects in enhancing the reversal of hepatic fibrosis and steatohepatitis occurring secondary to the induction with the diet that is deficient in methionine choline (Chen et al., 2015). Given that OEA has shown to have some therapeutic effect of modulating hepatic fibrosis by blocking the stimulation of the stellate cells in the hepatic system, it serves as an excellent potential treatment option for hepatic fibrosis in humans.
Other potent therapeutic agents with the potential to become some of the effective therapies for treating patients diagnosed with hepatic fibrosis include anticoagulants. Research designates that there is a link between hepatic fibrosis and some of the prothrombotic features, such as factor V Leiden (Nathwani et al., 2019). The exploration showed that mutations in the Leiden factor played a substantial function in tremendously increasing the rate of formation of hepatic fibrosis among patients who were suffering from hepatitis C infection. The activated forms of factor II and the Stuart–Prower factor also play a crucial part in the formation of LF by stimulating the stellate cells in the liver (Nathwani et al., 2019). An injury often results in the activation of the coagulation system. Subsequently, there is the formation of thrombin from the conversion of its precursor form, which is prothrombin. The two clotting factors then take part in stimulating the HSC using the protease receptors that are already active. One of the studies showed that mice that received rivaroxaban, which acts as an antagonist of Stuart–Prower factor, ended up developing a milder form of hepatic fibrosis compared to the control animal models despite their exposure to thioacetamide for at least two months (Dhar et al., 2018). Prolonged administration of low molecular heparin such as enoxaparin in animal models revealed an excellent regression of hepatic fibrosis, which would ultimately improve the existing portal hypertension. Fortunately, there is a small exploration carried out to determine the effectiveness of the use of heparin as part of the management regimens for hepatic fibrosis in humans. Individuals diagnosed with hepatitis B virus received heparin within a period of twenty-one days would reveal an improvement in their serums markers, especially ALT, while the concentrations of collagen type 4 reduced drastically (Nathwani et al., 2019). However, the long-term use of heparin in treating hepatic fibrosis may not be feasible due to its widespread adverse effects such as thrombocytopenia. As such, there is a prerequisite for more studies on humans to determine how anticoagulants can be useful in managing hepatic fibrosis, taking into consideration their adverse effects.
4.3.2.3 Blockers of HSC effects after stimulation
Antifibrotic agents also include drugs that inhibit the reaction of the stellate cells in the hepatic system after their activation. As such, the drugs aim at hindering the effect of the activated HSC from preventing the formation of the fibrotic tissue while reducing the chances of progression of fibrosis. One such group of drugs are the caspases inhibitors. Some studies indicate the administration of the caspase inhibitor IDN-6556 among people who underwent transplantation of the liver played a momentous function in preventing the risk of reperfusion injury (Woolbright et al., 2018). Such an injury would have triggered the pathways involved with inflammation, which would result in the activation of the myofibroblasts that would eventually lay off the matrix proteins in the hepatic system.
However, the effect of the inhibitor was only practical when the agent was included in the storage solution of the liver rather than the patient receiving it after the transplantation (Woolbright et al., 2018). In line with this, there was a concern that the administration of the caspase inhibitor following the transplantation might result in the blockage of action of other enzymes such as cathepsins secondary to the high amounts of the drug that led to most of the agents to reach off-target organs. When animal models such as mice with an induced form of hepatic fibrosis receive caspase inhibitors, they often reveal some protective effect against apoptosis, especially during the early stages following an injury. However, the effects tend to wane over time, with the cells starting to undergo necrosis barely one day after the initiation of the treatment with the caspase inhibitors. Thus, the findings demonstrate that in the short term, the caspase inhibitors might have a positive effect, but they are not safe for long-term use. Since some investigators still consider caspase inhibitors as some of the potential therapeutic agents for treating hepatic fibrosis, it is vital to note that LF is commonly a chronic condition and most of the underlying conditions contributing to the condition are also CLD such as NASH and alcohol-induced hepatitis (Woolbright et al., 2018). In such a case, the administration of caspase inhibitors would quickly result in causing more injury to the hepatic tissue, which indicates that its use in humans has more risks than benefits (Woolbright et al., 2018). As such, the study did not approve the utilization of the caspase inhibitors as part of the regimens for treating patients diagnosed with hepatic fibrosis due to its lack of specificity, which would otherwise result in a vast range of adverse effects. Researchers should take part in more explorations that aim at determining how to modify the caspase inhibitors in a way that its beneficial effects of protecting cells against apoptosis remain intact even in the long term when eradicating any chances of the agents causing hepatic injury.
Another therapeutic group of agents would be the drugs that work by antagonizing the role of endothelin such as bosentan. Even though most of the existing studies carried out on bosentan try to determine the role and its efficacy in managing patients suffering from pulmonary hypertension, some explorations indicate that it could possess some antifibrotic effect by working to block the stimulation of the stellate cells in the hepatic system (Nathwani et al., 2019). Among the major initial steps in the process of forming the LF is the instigation of the myofibroblasts from the stellate cells in the hepatic system. Subsequently, the cells secret a mixture of extracellular matrix proteins that go a long way in forming the fibrotic tissue in the liver. The HSC possesses an excellent number of endothelin receptors, which also play a crucial function during their activation process.
Following an injury to the hepatic tissue, the stellate cells migrate to the area to enhance the formation of the fibrotic tissue. Recent studies indicate that bosentan working through the endothelin receptors to antagonize their role can effectively block the stimulation of the stellate cells in the hepatic system, which would reduce the amount of matrix proteins secreted, resulting in a decline in the formation of hepatic fibrosis (Nathwani et al., 2019). Bosentan also acts directly by inhibiting the process of laying off of the extracellular matrix, which prevents the formation of fibrotic tissue in the liver (Nathwani et al., 2019). However, most studies exploring the function of bosentan in the management of people diagnosed with pulmonary hypertension indicate that the agent could have severe liver complications such as enhancing the progression of fibrosis to cirrhosis (Savale et al., 2013). As such, there is a requisite for more studies to determine whether bosentan can be an operational therapy for the management of LF among humans. Another therapeutic agent is cytosporone – B, which has the capability of blocking the TGF signalling pathway, which is one of the critical steps during the formation of fibrosis in the hepatic system (Nathwani et al., 2019). Among the primary aspect that enhances tissue repair is the responses of the mesenchymal cells. In case there is a failure to cause a halt into the process of tissue repair results in the establishment of fibrotic tissue, which affects the typical function of the organ affected. Currently, there are not many effective therapies that can enhance the inhibition of the formation of the fibrotic tissue in humans while restoring the normal functioning of the organs. A study carried out by Palumbo-Zerr and colleagues focused on exploring the effect of the nuclear receptor type 4AI gene agonists such as cytosporone, which essentially work by blocking the signaling pathways of the TGF-β that is imperative in the formation of fibrotic tissues in the liver (Palumbo-Zerr et al., 2015).
The nuclear receptor type 4A1 has the capability of recruiting other repressors whose primary role is to target the genes that code for TGF-β, enhancing its signaling route blockage. It is imperative to bear in mind that during a normal physiological process, there is a negative feedback loop between the two major pathways (Palumbo-Zerr et al., 2015). In relation to this, following an injury, there is the galvanization of the TGF-β signaling conduit to enhance wound healing, which in turn stimulates the expression of the nuclear receptor type 4A1, forming the feedback loop. However, under normal physiologic conditions, the continuous galvanization of the TGF-β signaling conduit eventually suppresses the expression and stimulation of the nuclear receptor type 4A1. Studies indicate that small quantities of agents that act as agonists of the latter can overcome the effects of the TGF-β motioning conduit resulting in blocking the process of fibrotic tissue formation in the hepatic and pulmonary systems in mice (Palumbo- Zerr et al., 2015).
In another exploration, the researchers aimed at determining whether cytosporone – B could be useful in the management of fibrosis affecting the vocal cords due to its antifibrotic effects. The results of the study confirmed cytosporone – B acting as an agonist of the nuclear receptor 4A1 gene reduced the expression of the genes coding for fibrosis whilst it reduced the stimulation of the TGF-β, which would otherwise have led to the galvanization of fibroblasts culminating in the deposition of the collagen and fibrotic tissue formation in the vocal cords (Hiwatashi et al., 2018). In connection with this, more explorations focusing on the nuclear receptor gene agonists such as cytosporone –B would be essential to determine its effectiveness in managing cases of hepatic fibrosis in humans. Other groups of drugs that work by blocking the reaction of the stellate cells in the hepatic system even after their activation are the therapeutic agents that target the farnesoid-X receptor. An excellent example of such drugs is obeticholic acid (Nathwani et al., 2019). Some prior explorations indicated that obeticholic acid was a fully effective treatment for hepatic fibrosis. However, the recent investigations claim that the therapeutic agent is not as effective in managing patients diagnosed with hepatic fibrosis since clinicians have to prescribe it in lower doses to avert the occurrence of its most commonly seen side effect, which is pruritus. A research carried out by Zhou and colleagues aimed at unveiling whether the combination of the obeticholic acid with other agents that act as blockers of apoptosis would effectively work towards treating patients with hepatic fibrosis (Zhou et al., 2019). During the study, a group of mice that had induced hepatic fibrosis received a combination of the obeticholic acid and the IDN-6556 that acts by inhibiting the process of apoptosis while the controls received only the obeticholic acid (Zhou et al., 2019). The outcomes of the exploration revealed that the mice which received both the IDN-6556 and a low dose of obeticholic acid expressed a less severe form of hepatic fibrosis since the obeticholic acid enhances the inhibition of stimulation of the stellate cells of the liver, which play a unique function in enhancing the formation of hepatic fibrosis (Zhou et al., 2019). As such, the combination therapy can offer some potential strategies for treating patients with hepatic fibrosis. The combination approach would be useful since the clinicians can give the obeticholic acid at a lower dose, which would avoid its side effects and still attain the same result of treating hepatic fibrosis.
Drugs that work via the inhibition of the ALK5, which serves as a receptor of the TGF-β, show some potential as an effective therapy for patients diagnosed with hepatic fibrosis (Nathwani et al., 2019). Among the major features of protracted forms of hepatic diseases is the accumulation of the matrix proteins, which is among the last steps, which results in the establishment of fibrotic tissue in the hepatic system. The TGF-β has been shown to have its effect of enhancing the process of fibrosis formation by acting via the ALK5 receptor through which it is capable of augmenting the transcription of the various genes that code for collagen types I and III (Baraut et al., 2015).
Among the most potent therapeutic agents that effectively block the action of the ALK5 receptor include GW6604 (Nathwani et al., 2019). An exploration carried out by Karkampouna and colleagues aimed at determining the effectiveness of the LY364947 in blocking the signaling corridors of TGF-β, mainly by blocking its type 1 receptor, which would eventually have an effect of reducing the formation of fibrotic tissue in the hepatic system, primarily focusing on animal models that had chronic liver diseases (Karkampouna et al., 2016). TGF-β plays a significant function in increasing the establishment of fibrosis. In addition to enhancing the expression of the genes that code for collagen, which enhances the formation of fibrosis, TGF-β also diminishes the process of breakdown of the matrix by hindering the action of metalloproteinases (Duarte et al., 2015). Studies also show that TGF-β may have a direct effect on the parenchymal cells, whereby its role is to block signals that enhance the proliferation of hepatocytes, especially after hepatectomy (Dewidar et al., 2019). In line with this, during cases of chronic forms of liver conditions, TGF-β plays a substantial part in preventing the normal repair process from taking place, which over time, results in the loss of hepatic function, principally through enhancing the formation of hepatic fibrosis. The outcomes of the exploration revealed that developing more drugs that could target to block the ALK5 receptor would be a possible means of acquiring an effective regimen for treating patients who develop hepatic fibrosis since the agents would prevent the deposition of the matrix. At the same time, they would augment the proliferation of hepatocytes making the liver to regain its normal function.
Drugs that target to inhibit the process, which results in the synthesis of collagen, are also potent therapeutic agents for LF (Nathwani et al., 2019). An excellent example of such drugs is halofuginone. The drug has the prospect to block the signaling conduit of the TGF, just like the ALK5 inhibitors. As such, it ensures that it curbs the production of the matrix while creating time for the hepatic cells to proliferate to ensure optimal function of the hepatic system during the drug administration. One of the studies carried out by Nelson and colleagues aimed at determining the antifibrotic effect of halofuginone in humans (Nelson et al., 2012). In line with this, there was the inoculation of halofuginone in the corneal fibroblasts derived from human donors. As such, the investigation was mainly in vitro. Subsequently, the corneal fibroblasts that had halofuginone would then receive TGF-beta, whose primary role was to try and stimulate the formation of fibrotic tissue. The use of western blots enabled the investigators to analyze the overall expression of fibronectin and the alpha-smooth muscles in the corneal fibroblasts. When focusing on the corneal tissue, the formation of the fibrotic tissue often culminates in a loss of vision for the victim. The transforming growth factor also plays a unique function in enhancing the formation of fibrosis, just like in the hepatic system (Nelson et al., 2012).
The results displayed that the inoculation of small quantities of halofuginone led to a momentous fall in the manifestation of genes coding for the matrix proteins, mostly the one coding for Smad3 (Nelson et al., 2012). In connection with this, there was a confirmation that halofuginone has an antifibrotic effect, which makes it a potential drug for treating patients diagnosed with hepatic fibrosis. Another exploration carried out by Parichatikanond and colleagues aimed at determining some of the agents that would be effective in the management of cardiac fibrosis (Parichatikanond et al., 2020). Following an injury to the cardiac muscle, there is the release of profibrotic elements such as endothelin-1, which in turn work to stimulate the cardiac fibroblasts, enhancing the deposition of matrix proteins. The establishment of fibrotic tissue in the heart, just like in the hepatic system, affects the normal functioning of the heart. As such, there is abnormal contractility of the cardiac muscles, and the remodeling process does not occur typically result in heart failure (Parichatikanond et al., 2020). In connection with this, drugs that inhibit the signaling pathway of TGF-β, such as halofuginone, would be potential treatment targets for cardiac fibrosis and myomas. Since halofuginone also works via various mechanisms such as blocking the production of matrix proteins directly and inhibiting the signaling pathway of TGF-beta, it would be an excellent treatment option for LF after further studies focusing on fibrosis occurring in the human hepatic system.
4.3.2.4 Enhancers of HSC apoptosis
Studies have also shown that other therapeutic agents that could have a beneficial effect in the management of patients who develop hepatic fibrosis are the drugs that mainly target to enhance the process of apoptosis among the activated stellate cells in the hepatic system (Nathwani et al., 2019). Gliotoxin is among such drugs. It was in the 1990s when one of the most widely renowned scientists and his colleagues brought up the notion that the process of formation of fibrotic tissue in the hepatic system is reversible (Shafigullina et al., 2018). It is the notion that has led many investigators in the medical field to take part in exploring some of the antifibrotic therapies that can enhance the management of hepatic fibrosis. The recent advancements when it comes to unveiling the exact pathophysiology of hepatic fibrosis enhanced the same process. In line with this, the primary cells that take part in determining the proteins from the extracellular matrix include the myofibroblasts, which mainly come from diverse sources such as the stellate cells of the hepatic system of the fibroblasts in the portal system. One of the explorations carried out by Shafigullina and colleagues aimed at determining whether Gliotoxin had a role in enhancing apoptosis among the stellate cells of the liver, especially after their activation (Shafigullina et al., 2018).
Gliotoxin has been shown to possess the potential of stimulating the process of apoptosis of macrophages and thymocytes. Even though prior research by Young-Oh Kweon and colleagues revealed that Gliotoxin had an effect that could stimulate apoptosis of HSC, the exploration never explained the exact mechanism through which it achieves the stimulation (Shafigullina et al., 2018). Studies indicate that Gliotoxin stimulates the apoptosis of the activated stellate cells of the hepatic system while decreasing the formation of the fibrotic tissue in the liver (Shafigullina et al., 2018). As such, Gliotoxin can be potential as an effective therapy for managing individuals diagnosed with hepatic fibrosis if there were other studies carried out using human subjects.
Another group of therapeutic agents would be the drugs that target the action of the NF-κB (Nathwani et al., 2019). NF-κB is among the most significant factors that play a unique purpose in facilitating the inflammatory reaction. The transcription factor works by mainly acting via the signaling conduits involved in inflammatory reactions. The stimulation of the NF-κB occurs in almost every chronic form of a hepatic condition such as NASH and alcoholic hepatic conditions (Luedde & Schwabe, 2011). The galvanization of the NF-κB mainly occurs secondary to any condition that creates a sense of potential danger resulting in the stimulation of an inflammatory or wound healing process. In line with this, the stimulation mainly happens through molecules derived from pathogens that act via the toll-like receptors and the inflammatory cytokines such as interleukin 1 (Luedde & Schwabe, 2011). Subsequently, the NF-κB plays a decisive function in affecting the survival of the hepatocytes and the stimulation of the stellate cells in the hepatic system. NF-κB enhances the proinflammatory responses and other reaction that prevents programmed cell death, especially protecting the hepatocytes while still ensuring that there are proper inflammatory and immune reactions when required (Luedde & Schwabe, 2011). In line with this, therapeutic agents such as sulfasalazine and thalidomide that target the NF-κB pathways inhibiting the signaling processes result in hindering the continuation of the inflammatory reaction, which would otherwise enhance the formation of fibrotic tissue in the hepatic system (Nathwani et al., 2019). Both agents play an influential role in suppressing chronic hepatic inflammatory reactions, making them some of the potent drugs to be effective as part of the regimens for treating patients with LF. However, there is a need to determine how such agents can be useful, bearing in mind that thalidomide has some teratogenicity adverse effects.
Another group of therapeutic agents are the hepatocyte growth factor (HGF). One of the explorations carried out in the early 1980s indicated that hepatocytes derived from adult rats would undergo through the process of proliferation once put in a culture that contained EGF and insulin, instilling the need among researchers to explore more on agents that could have hepatotropic effects (Nakamura & Mizuno, 2010).
Further studies would then reveal the presence of a putative factor that had the capability of enhancing the stimulation of the production of DNA, which would eventually cause the hepatocytes to proliferate in the animal models (Nakamura & Mizuno, 2010). The factor would come to be known as the HGF. One of the researches carried out by Zhang Yichi and colleagues aimed at unveiling whether HGF had a therapeutic effect in a liver that had cirrhosis induced using carbon tetrachloride in an animal model (Zhang et al., 2018). In connection with this, mesenchymal cells of the bone marrow among the mice underwent some labeling to make them overexpress HGF. The investigators would then transfer the mesenchymal cells that had overexpression of HGF to the rats, which had hepatic cirrhosis using the intravenous route. The study would then progress for one month (Zhang et al., 2018). The outcomes of the investigation indicated that the mice that received the mesenchymal cells expressing excessive amounts of HGF showed significant regression in their hepatic cirrhosis when likened to the control group that never received the mesenchymal cells. The serum quantities of ALT and AST also showed a tremendous decline among the rats that received the mesenchymal cells illustrating the fact that HGF could have a potential of protecting the liver from further injury, which otherwise contributes in the rising levels of the transaminases (Zhang et al., 2018). Studies also indicate that the mesenchymal cells have the potential of undergoing differentiation and become hepatocytes meaning that they have the capability of enhancing hepatic regeneration (Zhang et al., 2018). In connection with this, the HGF could have a positive effect on augmenting the recovery of the hepatic tissue. As such, further explorations using human models would be imperative to determine the effectiveness of the use of hepatic growth factor as potential therapeutic agents for treating patients with hepatic fibrosis.
Other potential agents for treating patients with hepatic fibrosis include interferon. Cytokines and growth factors are among the chief players in the formation of fibrotic tissue in the hepatic system. Interferon-gamma secreted by the natural killer cells in the fibrotic tissue in the hepatic system has the function of enhancing antifibrotic activities. As such, most researchers explore the possible strategies of utilizing the cytokine as part of the agents that can effectively manage hepatic fibrosis (van Dijk et al., 2015). However, most trials demonstrated widespread adverse effects after the administration of interferon, and the agent had low efficacy (van Dijk et al., 2015). One of the explorations carried out by van Dijk Fransien and colleagues demonstrated that the modified form of interferon in combination with a growth factor- BB resulting from platelets might have the potential of becoming useful for managing hepatic fibrosis (van Dijk et al., 2015). According to Nathwani et al. (2019), interferon work by stimulating the effect of natural killer cells and promotes programmed cell death of stellate cells in the liver, which indicates that it can be useful for reducing the formation of fibrotic tissue in the hepatic system.
4.4. Conclusion
This section majored in analyzing data from numerous journal articles that are peer-reviewed to acquire data on some of the chief developments of therapeutic agents that would be effective in the management of patients who develop hepatic fibrosis. Some of the articles reviewed were systematic reviews, while others were empirical explorations. It is fundamental to bear in mind that only several explorations exist that focus on the use of therapeutic agents that can be effective in managing people diagnosed with hepatic fibrosis. Most of these trials claim that some of the agents might not be effective for human use due to the widespread adverse effects. Most findings of the explorations demonstrate the need to have a full appreciation of the molecular pathways that contribute to LF since that is the easiest way to develop therapeutic agents effective against hepatic fibrosis.
Chapter 5: Conclusion
5.1 Introduction
The exploration aimed at presenting some chief information about the major potential therapeutic agents that target to treat patients diagnosed with hepatic fibrosis. Chapter one provided a vast range of background related to the topic, including some of the critical pathogenic pathways which act as the primary targets for most of the potential therapeutic agents for hepatic fibrosis. The initial chapter also provided the aims and primary objectives of the investigation, which would guide the development of the subsequent chapters. This segment will offer an overview of the chief verdicts of the exploration and some of the recommendations that would enhance the area of managing hepatic fibrosis. The last part of this section will be a conclusion that offers a brief deduction of this investigation.
5.2 Main Findings
Currently, the most effective way of treating patients who develop hepatic fibrosis is by eradicating the underlying disease. For example, in case the cause of the hepatic fibrosis is viral hepatitis, patients should receive the appropriate antiviral drugs to suppress the viral load, which would eventually enhance the reversal of hepatic fibrosis. In case the primary contributor to hepatic fibrosis is alcohol consumption, the initial step should be the avoidance of taking alcohol. Understanding the central pathways that lead to the pathogenesis of LF is among the critical steps of unveiling novel therapeutic agents with the potential to treat the condition effectively. Since such knowledge has been on the rise due to advancements in the field of science, it contributes to more researchers working on some of the potential agents that could be effective in managing patient’s diagnosed hepatic fibrosis. Unfortunately, most existing studies that demonstrate some agents that could effectively manage hepatic fibrosis used animal models as the sample population.
The few that utilize animal models illustrate the use of most of such agents results in widespread side effects, thus not fully approving their use among humans. Some of the agents shown to have the potential to treat hepatic fibrosis effectively falls into four major categories. The first category includes agents that aim at blocking the pathways that lead to inflammatory and immune reactions. Examples of medications that fall under this group include caffeine, glucocorticoids, and curcumin (Nathwani et al., 2019). Another group of agents the ones that focus on targeting the already activated stellate cells of the liver, aiming at blocking them from functioning. Such agents include vitamin E and pioglitazone (Nathwani et al., 2019). The third category of therapeutic agents aims at blocking the effects that would otherwise manifest after the galvanization of the stellate cells in the hepatic system. Examples of such drugs include bosentan and obeticholic acid. The last group of potential antifibrotic agents is the one that works by stimulating the apoptosis of the already activates stellate cells in the liver. Examples of drugs in this group include Gliotoxin and interferon (Nathwani et al., 2019).
5.3 Recommendations
Among an essential factor to consider is the fact that most of the existing explorations involved animal models. Therefore, there is a need to take part in investigations that will involve human models to determine the effectiveness of most of the therapeutic agents that show potential effectiveness in treating hepatic fibrosis among animal models. Researchers should consider the integration of more nanoparticles when developing the drugs that target treating hepatic fibrosis in humans to reduce their adverse effects. Further studies are obligatory to define the efficacy of most of the potential antifibrotic agents in the clinical field. Researchers have to explore the possibility of combination therapies as a means of reducing the adverse effects seen with the use of high doses of some potential therapeutic agents against hepatic fibrosis. The treatment of patients who develop hepatic fibrosis must remain individualized to enhance the success of the treatment.
5.4: Conclusion
Understanding the pathogenesis of hepatic fibrosis is paramount in exploring that various drugs which have the potential of treating the condition. Numerous studies exist that focus on unveiling drugs that halt or enhance the reversal of hepatic fibrosis. However, there is a need for further studies, mainly focusing on human models to ascertain the most effective therapeutic agents in humans. At present, the most effective form of treatment for hepatic fibrosis remains the eradication of the underlying cause.
References:
- Abutabenjeh, S. & Jaradat, R., 2018. Clarification of research design, research methods, and research methodology: A guide for public administration researchers and practitioners. Teaching Public Administration, 36(3), p. 237–58.
- Afdhal, N. H., 2012. Fibroscan (transient elastography) for the measurement of liver fibrosis. Gastroenterology & Hepatology, 8(9), pp. 605-7.
- Ahmad, W. I. B. G. S. A. S. K. S. et al., 2011. A brief review on molecular, genetic, and imaging techniques for HCV fibrosis evaluation. Virology Journal, 8(8),p. 53.
- Altamirano-Barrera, A., Barranco-Fragoso, B. & Méndez-Sánchez, N., 2017. Management strategies for liver fibrosis. Annals of Hepatology, 16(1), pp. 48-56.
- Ambade, A. et al., 2019. Pharmacological inhibition of ccr2/5 signaling prevents and reverses alcohol-induced liver damage, steatosis, and inflammation in mice. Hepatology, 69(3), p. 1105–1121.
- Aravalli, R. N. & Steer, C. J., 2017. Immune-mediated therapies for liver cancer. Genes, 8(2), p. 76.
- Baeck, C., Wei, X. & Bartneck, M., 2014. Pharmacological inhibition of the chemokine C-C motif chemokine ligand 2 (monocyte chemoattractant protein 1) accelerates liver fibrosis regression by suppressing Ly-6C (+) macrophage infiltration in mice. Hepatology,59(3), pp. 1060-72.
- Bajaj, J. S. et al., 2011. The multi-dimensional burden of cirrhosis and hepatic encephalopathy on patients and caregivers. American Journal of Gastroenterology, 106(9), pp. 1646-53.
- Bansal, R., Nagórniewicz, B. & Prakash, J., 2016. Clinicaladvancements in the targeted therapies against liver fibrosis. Mediators of Inflammation, Volume 2016, p. 7629724. Bansal, R. et al., 2011. Novel engineered targeted interferon-gamma blocks hepatic fibrogenesis in mice. Hepatology, 54(2), pp. 586-96.
- Baraut, J. et al., 2015. Transforming growth factor-β increases interleukin-13 synthesis via GATA-3 transcription factor in T-lymphocytes from patients with systemic sclerosis. Arthritis Research & Therapy, 17(196), pp. 1-15.
- Bartneck, M., Warzecha, K. T. & Tacke, F., 2014. Therapeutic targeting of liver inflammation and fibrosis by nanomedicine. Hepatobiliary Surgery and Nutrition, 3(6), p. 364–76.
- Berres, M. et al., 2010. Antagonism of the chemokine Ccl5 ameliorates experimental liver fibrosis in mice. J. The Journal of Clinical Investigation, 120(11), pp. 4129-40.
- Beste, L. A. et al., 2015. Trends in burden of cirrhosis and hepatocellular carcinoma by underlying liver disease in US veterans, 2001-2013. Gastroenterology, 149(6), pp. 1471-1482.e5.
- Bian, Z. &Ma, X., 2012. Liver fibrogenesis in non-alcoholic steatohepatitis. Frontiers in Physiology, Volume 3, p. 248.
- Boettcher, E. et al., 2012. Meta-analysis: Pioglitazone improves liver histology and fibrosis in patients with non-alcoholic steatohepatitis. Alimentary Pharmacology & Therapeutics, 35(1), pp. 66-75.
- Brenner, D. A. et al., 2012. Origin of myofibroblasts in liver fibrosis. Fibrogenesis & Tissue Repair, 5(Suppl 1), p. S17.
- Campbell, R., Pound, P. & Morgan, M., 2011. Evaluating meta-ethnography: Systematic analysis and synthesis of qualitative research. Health Technology Assessment, 15(43), pp. 1-164.
- Castera, L. & Pinzani, M., 2010. Biopsy and non-invasive methods for the diagnosis of liver fibrosis: Does it take two to tango? Gut, 59(7), pp. 861-66.
- Chang, E., Park, C. Y. & Park, S. W., 2013. Role of thiazolidinediones, insulin sensitizers, in non-alcoholic fatty liver disease.Journal of Diabetes Investigation, 4(6), pp. 517-24.
- Cheng, J. Y.-K. & Wong, G. L.-H., 2017. Advances in the diagnosis and treatment of liver fibrosis. Hepatoma Research, Volume 3, pp. 156-69.
- Cheng, J. Y. et al., 2016. Metabolic syndrome increases cardiovascular events but not hepatic events and death in patients with chronic hepatitis B. Hepatology, 64(5), pp. 1507-17.
- Chen, L. et al., 2015. Oleoylethanolamide, an endogenous PPAR-α ligand, attenuates liver fibrosis targeting hepatic stellate cells. Oncotarget,6(40), p. 42530–40.
- Cinar, R. et al., 2016. Hybrid inhibitor of peripheral cannabinoid-1 receptors and inducible nitric oxide synthase mitigates liver fibrosis. JCI Insight, 1(11), p. e87336. Cooper, C. et al., 2018. Defining the process to literature searching in systematic reviews: A literature review of guidance and supporting studies. BMC Medical Research Methodology, 18(1), p. 85.
- D’Ambrosio, R., Aghemo, A. & Rumi, M., 2012. A morphometric and immunohistochemical study to assess the benefit of sustained virological response in hepatitis C virus patients with cirrhosis. Hepatology, 56(2), pp. 532-43.
- Dewidar, B., Meyer, C., Dooley, S. & Meindl-Beinker, A. N., 2019. TGF-β in hepatic stellate cell activation and liver fibrogenesis-updated 2019.Cells, 8(11), p. 1419.
- Dhar, A. et al., 2018. Thrombin and factor Xa link the coagulation system with liver fibrosis.BMC Gastroenterology, 18(1),p. 60.
- Dibba, P. et al., 2018. Mechanistic potential and therapeutic implications of cannabinoids in non-alcoholic fatty liver disease. Medicines, 5(2), p. 47.
- Duarte, S., Baber, J., Fujii, T. & Coito, A. J., 2015. Matrix metalloproteinases in liver injury, repair, and fibrosis.Matrix Biology, Volume 44-46, p. 147–56.
- Ebrahimi, H., Naderian, M. & Sohrabpour, A.A., 2016. New concepts on pathogenesis and diagnosis of liver fibrosis; a review article. Middle East Journal of Digestive Diseases, 8(3), p. 166–78.
- Estes, C. et al., 2018. Modeling the epidemic of non-alcoholic fatty liver disease demonstrates an exponential increase in the burden of disease. Hepatology, 67(1), p. 123– 33.
- Forner, A., Llovet,J. & Bruix, J., 2012. Hepatocellular carcinoma. The Lancet, 379(9822), pp. 1245-55.
- Giby, V. G. & Ajith, T. A., 2014. Role of adipokines and peroxisome proliferator-activated receptors in non-alcoholic fatty liver disease. World Journal of Hepatology, 6(8), p. 570–79.
- Gutiérrez-Grobe, Y. et al., 2012. High coffee intake is associated with lower grade non-alcoholic fatty liver disease: The role of peripheral antioxidant activity. Annals of Hepatology, 11(3), pp. 350-55.
- He, L., Liu, X., Wang, L. & Yang, Z., 2016. Thiazolidinediones for nonalcoholic steatohepatitis: A meta-analysis of randomized clinical trials. Medicine, 95(42), p. e4947.
- Hernández, J. A., López-Sánchez, R. C. & Rendón-Ramírez, A., 2016. Lipids and oxidative stress associated with ethanol-induced neurological damage. OxidativeMedicine and CellularLongevity, Volume 2016, p. 1543809.
- Hiwatashi, N., Mukudai, S., Bing, R. & Branski,R. C., 2018. The effects of cytosporone-B, a novel antifibrotic agent, on vocal fold fibroblasts. The Laryngoscope, 128(12), pp. E425-E428.
- Huang, C. & Ogawa, R., 2012. Fibroproliferative disorders and their mechanobiology.
- Connective Tissue Research,53(3), pp. 187-96.
- Huang, Y., Deng, X. & Liang, J., 2017. Modulation of hepatic stellate cells and reversibility of hepatic fibrosis.Experimental Cell Research, 352(2), pp. 420-26.
- Jakobsen, J. C. et al., 2017. Direct-acting antivirals for chronic hepatitis C. Cochrane Library: Cochrane Reviews, 6(6), p. CD012143.
- Jeong, C. H. & Joo, S. H., 2016. Downregulation of reactive oxygen species in apoptosis. Journal of Cancer Prevention, 21(1), p. 13–20.
- Jiang, J. X. et al., 2010. NOX2 plays a key role in stellate cell activation and liver fibrogenesis in vivo. Gastroenterology, 139(4), p. 1375–1384.e4.
- Jourdan, T. et al., 2013. Activation of the Nlrp3 inflammasome in infiltrating macrophages by endocannabinoids mediates beta cell loss in type 2 diabetes. Nature Medicine,19(9), pp. 1132-40.
- Julien, J. et al., 2020. Projected prevalence and mortality associated with alcohol-related liver disease in the USA, 2019–40:A modeling study. The Lancet, 5(6),p. e316– 23.
- Karkampouna, S. et al., 2016. Inhibition of TGFβ type I receptor activity facilitates liver regeneration upon acute CCl4 intoxication in mice. Archives of Toxicology, 90(2), pp. 347-57.
- Karlmark, K., Zimmermann, H. & Roderburg, C., 2010. The fractalkine receptor CX₃CR1 protects against liver fibrosis by controlling differentiation and survival of infiltrating hepatic monocytes. Hepatology,52(5), pp. 1769-82.
- Karsdal, M. A. et al., 2015. Novel insights into the function and dynamics of extracellular matrix in liver fibrosis. American Journal of Physiology-Gastrointestinal and Liver Physiology, 308 (10), p. G807–G830.
- Kim, M. et al., 2012. Beneficial effects of candesartan, an angiotensin-blocking agent, on compensated alcoholic liver fibrosis – A randomized open-label controlled study. Liver International, 32(6), pp. 977-87.
- Knop, V. H. D. et al., 2016. Regression of fibrosis and portal hypertension in HCV- associated cirrhosis and sustained virologic response after interferon-free antiviral therapy. Journal of Viral Hepatitis, 23(12), pp. 994-1002.
- Kong, X., Feng, D., Mathews, S. & Gao, B., 2013. Hepatoprotective and anti-fibrotic functions of interleukin-22: Therapeutic potential for the treatment of alcoholic liver disease. Journal of Gastroenterology & Hepatology, 28(01), pp. 50-60.
- Lee, U. E. & Friedman, S. L., 2011.Mechanisms of hepatic fibrogenesis. Best Practice & Research: ClinicalGastroenterology, 25(2), p. 195–206.
- Lee, Y. A., Wallace, M. C. & Friedman, S. L., 2015. Pathobiology of liver fibrosis: A translational success story. Gut, 64(5), p. 830–41.
- Lefebvre, E. et al., 2016. Antifibrotic effects of the dual CCR2/CCR5 antagonist cenicriviroc in animal models of liver and kidney fibrosis. PloS One, 11(6), p. e0158156.
- Li, H., You, H., Fan, X. & Jia, J., 2016. Hepatic macrophages in liver fibrosis: pathogenesis and potential therapeutic targets. BMJ Open Gastroenterology, 3(1), p. e000079.
- Li, J. et al., 2012. Plasmasphingolipids: Potential biomarkers for severe hepatic fibrosis in chronic hepatitis C. Molecular Medicine Reports, 12(1), 323-30.
- Liu, T. et al., 2012. Molecularserum markers of liver fibrosis. Biomarker Insights, Volume 7, p. 105–17.
- Loomba, R. et al., 2018. The ASK1 inhibitor selonsertib in patients with nonalcoholic steatohepatitis: A randomized, phase 2 trial.Hepatology, 67(2), pp. 549-59.
- Love, K. T. et al., 2010. Lipid-like materials for low-dose, in vivo gene silencing. Proceedings of the National Academy of Sciences of the United States of America, 107(5), p. 1864–69.
- Luedde, T. & Schwabe, R. F., 2011. NF-κB in the liver–linking injury, fibrosis, and hepatocellular carcinoma. Nature Reviews Gastroenterology & Hepatology, 8(2), p. 108–18.
- Lurie, Y. et al., 2015. Non-invasive diagnosis of liver fibrosis and cirrhosis. World Journal of Gastroenterology, 21(41), p. 11567–83.
- Mahady, S. E. et al., 2011. The role of thiazolidinediones in non-alcoholic steatohepatitis – A systematic review and meta-analysis. Journal of Hepatology, 55(6), pp. 1383-90.
- Mallett, R., Hagen-Zanker, J. & Maren, R. S., 2012. The benefits and challenges of using systematic reviews in international development research. Journal of Development Effectiveness, 4(3), pp. 445-55.
- Marcellin, P., Gane, E. & Buti, M., 2013. Regression of cirrhosis during treatment with tenofovir disoproxil fumarate for chronic hepatitis B: A 5-year open-label follow-up study. Lancet, 381(9865), pp. 468-75.
- Mellinger, J., Shedden, K. & Winder, G., 2018. The high burden of alcoholic cirrhosis in privately insured persons in the United States.Hepatology, 68(3), pp. 872-82.
- Modi, A. A. et al., 2010. Increased caffeine consumption is associated with reduced hepatic fibrosis. Hepatology, 51(1), pp. 201-9.
- Montano-Loza, A., Thandassery, R. & Czaja, A., 2016. Targeting hepatic fibrosis in autoimmune hepatitis. Digestive Diseases and Sciences, 61(11), pp. 3118-139.
- Moon, A. M., Green, P. K., Berry, K. & Ioannou, G. N., 2017. Transformation of hepatitis C antiviral treatment in a national healthcare system following the introduction of direct antiviralagents. Alimentary Pharmacology & Therapeutics, 45(9), pp. 1201-121.
- Moon, H. J., Finney, J., Ronnebaum, T. & Mure, M., 2014. Human lysyl oxidase-like 2. Bioorganic Chemistry, Volume 57, pp. 231-41.
- Mudaliar, S. et al., 2013. Efficacy and safety of the farnesoid X receptor agonist obeticholic acid in patients with type 2 diabetes and non-alcoholic fatty liver disease. Gastroenterology, 145(3), pp. 574-82.e1.
- Nakamura, T. & Mizuno, S., 2010. The discovery of hepatocyte growth factor (HGF) and its significance for cell biology, life sciences, and clinical medicine. Proceedings of the Japan Academy. Series B, Physical and Biological Sciences,88(6), pp. 588-610.Nathwani, R. et al., 2019. A review of liver fibrosis and emerging therapies. European Medical Journal, 4(4), pp. 105-16.
- Navarro, L. A. et al., 2015. Arginase 2 deficiency result sin spontaneous steatohepatitis: A novel link between innate immune activation and hepatic de novo lipogenesis. Journal of Hepatology, 62(2), pp. 412-20.
- Nelson, E. F. et al., 2012.Halofuginone down-regulates Smad3expression and inhibits the TGFbeta-induced expression of fibrotic markers in human corneal fibroblasts. Molecular Vision, Volume18, p. 479–87.
- Neuschwander-Tetri, B. A. et al., 2015. Farnesoid X nuclear receptor ligand obeticholic acid for non-cirrhotic, non-alcoholic steatohepatitis (FLINT): A multicentre, randomised, placebo-controlled trial. Lancet, 385(9972), pp. 956-65.
- Nevens, F. et al., 2016. A placebo-controlled trial of obeticholic acid in primary biliary cholangitis. The New England Journal of Medicine, 375(7), pp. 631-43. Ogawa, S., Ochi, T. & Shimada, H., 2010. Anti-PDGF-B monoclonal antibody reduces liver fibrosis development. Hepatology Research, 40(11), pp. 1128-41.
- Okimoto, S. et al., 2019. Vitamin A-coupled liposomal Rho-kinase inhibitor ameliorates liver fibrosis without systemic adverse effects. Hepatology Research, 49(6), pp. 663-75.
- Osna, N. A., Donohue, T. M. & Kharbanda, K. K., 2017. Alcoholic liver disease: Pathogenesis and current management. Alcohol Research: Current Reviews, 38(2),p. 147–61.
- Ovacik, M. & Lin, K., 2018. Tutorial on monoclonal antibody pharmacokinetics and its considerations in early development. Clinical and Translational Science, 11(6), pp. 540-52.
- Palumbo-Zerr, K. et al., 2015. Orphan nuclear receptor NR4A1 regulates transforming growth factor-β signaling and fibrosis. Natural Medicine, 21(2), pp. 150-58.
- Parichatikanond, W., Luangmonkong, T., Mangmool, S. & Kurose,H., 2020. Therapeutic targets for the treatment of cardiac fibrosis and cancer: Focusing on TGF-β signaling. Frontiers in Cardiovascular Medicine, Volume 7, p. 34.
- Parikh, N., Marrero, W. & Wang, J., 2019. Projected increase in obesity and non- alcoholic-steatohepatitis-related liver transplantation waitlist additions in the United States. Hepatology, 70(2), pp. 487-95.
- Pasquetto, M. et al., 2011. Targeted drug delivery using immunoconjugates: Principles and applications. Journal of Immunotherapy, 34(9), pp. 611-28.
- Patel, A. A. et al., 2017. Palliative care and health care utilization for patients with end-stage liver disease at the end of life.Clinical Gastroenterology and Hepatology, 15(10), pp. 1612-1619.e4.
- Petitclerc, L., Gilbert, G., Nguyen, B. N. & Tang,A., 2017. Liver fibrosis quantification by Magnetic Resonance Imaging. Tropics in Magnetic Resonance Imaging, 26(6), p. 229–41.
- Philips, G. M. et al., 2011. Hedgehog signaling antagonist promotes regression of both liver fibrosis and hepatocellular carcinoma in a murine model of primary liver cancer. PloS One, 6(9), p. e23943.
- Pines, M., 2014. Halofuginone for fibrosis, regeneration, and cancer in the gastrointestinal tract. World Journal of Gastroenterology, 20(40), p. 14778–86.
- Poilil Surendran, S., George Thomas,R., Moon, M. J. & Jeong, Y. Y., 2017. Nanoparticles for the treatment of liver fibrosis. International Journal of Nanomedicine, Volume12, p. 6997–7006.
- Poynard, T. et al., 2011. Prognostic value of liver fibrosis biomarkers: A meta-analysis. Gastroenterology & Hepatology, 7(7), pp. 445-54.
- Puche, J. E. et al., 2013. A novel murine model to deplete hepatic stellate cells uncovers their role in amplifying liver damage in mice. Hepatology, 57(1), pp. 339-50.
- Puche, J., Saiman, Y. & Friedman,S., 2013. Hepatic stellate cells and liver fibrosis.
- Comprehensive Physiology, 3(4), pp. 1473-92.
- Qu, K. et al., 2015. New insight into the anti-liver fibrosis effect of multitargeted tyrosine kinase inhibitors: From molecular target to clinical trials. Frontiers in Pharmacology, Volume 6, p. 300.
- Redza-Dutord, M. & Averill-Bates, D. A., 2016. Activation of apoptosis signaling pathways by reactive oxygen species. Biochimica et Biophysica Acta, Volume 2016, pp. 2977-92.
- Rein, D. B. et al., 2012. The cost-effectiveness of birth-cohort screening for hepatitis C antibody in U.S. primary care settings. Annals of Internal Medicine, 156(4), pp. 263- 70.
- Robinson, P. & Lowe, J., 2015. Literature reviews and systematic reviews. Australian and New Zealand Journal of Public Health, 39(2), p. 103.
- Sahin, H., Borkham-Kamphorst, E. & Kuppe, C., 2012. Chemokine Cxcl9 attenuates liver fibrosis-associated angiogenesis in mice. Hepatology, 55(5), pp. 1610-19.
- Saiman, Y. & Friedman,S. L., 2012. The role of chemokines in acute liver injury. Frontiers in Physiology, 3(213), pp. 1-12.
- Sánchez-Valle, V., Chávez-Tapia, N., Uribe, M. & Méndez-Sánchez, N., 2012. Role of oxidative stress and molecular changes in liver fibrosis: A review. Current Medicinal Chemistry, 19(28), pp. 4850-60.
- Sanyal, A. J. et al., 2010. Pioglitazone, vitamin E, or placebo for non-alcoholic steatohepatitis. The New England Journal of Medicine, 362(18), p. 1675–85.
- Savale, L. et al., 2013. Efficacy, safety, and pharmacokinetics of bosentan in portopulmonary hypertension. European Respiratory Journal, 41(1), pp. 96-103.
- Schiff, E. R. et al., 2011. Long-term treatment with entecavir induces reversal of advanced fibrosis or cirrhosis in patients with chronic hepatitis B. Clinical
- Gastroenterology and Hepatology, 9(3), pp. 274-276.e1.
- Schuppan, D., 2015. Liver fibrosis: Common mechanisms and antifibrotic therapies. Clinics and Research in Hepatology and Gastroenterology, 39(Suppl 1), p. S51–S59.Schuppan, D. & Kim, Y. O., 2013. Evolving therapies for liver fibrosis. The Journal of Clinical Investigation, 123(5), pp. 1887-1901.
- Sebastiani, G., Gkouvatsos, K. & Pantopoulos, K., 2014. Chronic hepatitis C and liver fibrosis. World Journal of Gastroenterology, 20(32), p. 11033–53.
- Seers, K., 2015. Qualitative systematic reviews: Their importance for our understanding of research relevant to pain. British Journal of Pain, 9(1), p. 36–40.
- Shafigullina, A. et al., 2018. Effect of curcumin and Gliotoxin on rat liver myofibroblast culture. Bio NanoScience, Volume8, pp. 522-36.
- Silvestri, C. & Di Marzo,V., 2013. The endocannabinoid system in energy homeostasis and the etiopathology of metabolic disorders. Cell Metabolism, 17(4), pp. 475-90.
- Singh, S. et al., 2015. Comparative effectiveness of pharmacological interventions for nonalcoholic steatohepatitis: A systematic review and network meta-analysis. Hepatology,62(5), pp. 1417-32.
- Stockmann, C. et al., 2010. Loss of myeloid cell-derived vascular endothelial growth factor accelerates fibrosis. Proceedings of the National Academy of Sciences of the United States of America, 107(9), p. 4329–34.
- Suk, K. T., & Kim, D. J., 2015. Staging of liver fibrosis or cirrhosis: The role of hepatic venous pressure gradient measurement. World Journal of Hepatology, 7(3),607–15.
- Syn, W. K. et al., 2012. NKT-associated hedgehog and osteopontin drive fibrogenesis in non-alcoholic fatty liver disease. Gut, 61(9), p. 1323–29.
- Tacke, F. & Trautwein, C., 2015. Mechanisms of liver fibrosis resolution. Journal of Hepatology, 63(4), pp. 1038-39.
- Tanaka, M. & Miyajima, A., 2016. Liver regeneration and fibrosis after inflammation. Inflamm Regener,36(1), pp. 1-6.
- Tapper, E. B. et al., 2015. Standard assessments of frailty are validated predictors of mortality in hospitalized patients with cirrhosis. Hepatology, 62(2), pp. 584-90.
- Tapper, E. B. & Parikh, N. D., 2018. Mortality due to cirrhosis and liver cancer in the United States,1999-2016: Observational study.The BMJ, Volume 362,p. k2817.
- Tapper, E., Catana, A. & Sethi, N., 2016. Direct costs of care for hepatocellular carcinoma in patients with hepatitis C cirrhosis. Cancer, 122(6), pp. 852-58.
- Tapper, E. et al., 2015. Cost-effective evaluation of non-alcoholic fatty liver disease with NAFLD fibrosis score and vibration controlled transient elastography. American Journal of Gastroenterology, 110(9), pp. 1298-304.
- Teschke, R., 2018. Alcoholic liver disease: Alcohol metabolism, cascade of molecular mechanisms, cellular targets, and clinical aspects. Biomedicines, 6(4), p. 106.
- Thabut, D. et al., 2011. Complementary vascular and matrix regulatory pathways underlie the beneficial mechanism of action of sorafenib in liver fibrosis. Hepatology, 54(2), p. 573–85.
- Thabut, D. & Shah, V., 2010. Intrahepatic angiogenesis and sinusoidal remodeling in chronic liver disease: New targets for the treatment of portal hypertension. Journal of Hepatology, 53(5), pp. 976-80.
- Toye, F., Seers, K. & Allcock, N., 2013. A meta-ethnography of patients’ experience of chronic non-malignant musculoskeletal pain. Health Services and Delivery Research, 1(12).
- Traber, P. G. et al., 2013. Regression of fibrosis and reversal of cirrhosis in rats by galectin inhibitors in thioacetamide-induced liver disease. PloS One, 8(10), p. e75361.
- Trautwein, C., Friedman, S., Schuppan, D. & Pinzani, M., 2015. Hepatic fibrosis: Concept to treatment. Journal of Hepatology, 62(1Suppl), pp. S15-S24.
- Udompap, P., Kim, D. & Kim, W. R., 2015. Current and future burden of chronic nonmalignant liver disease. Clinical Gastroenterology and Hepatology, 13(12), p. 2031–204.
- van Dijk, F., Olinga, P., Poelstra, K. & Beljaars, L., 2015. Targeted therapies in liver fibrosis: Combining the best parts of platelet-derived growth factor-BB and interferon-gamma. Frontiers in Medicine, Volume 2, p.72.
- Vilar-Gomez, E. et al., 2015. Weight loss through lifestyle modification significantly reduces features of non-alcoholic steatohepatitis. Gastroenterology, 149(2), pp. 367-78.Wang, H., Naghavi, M. & Allen, C., 2016. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980-2015: A systematic analysis for the global burden of disease study 2015. Lancet, 388(10053), pp. 1459-544.
- Wang, P. et al., 2016. Promising therapy candidates for liver fibrosis. Frontiers in Physiology, 7(47), pp. 1-9.
- Wasmuth, H., Tacke,F. & Trautwein, C., 2010. Chemokines in liver inflammation and fibrosis. Seminars in Liver Disease, 30(3), pp. 215-25.
- Wee, B. V. & Banister, D., 2016. How to write a literature review paper. Transport Reviews, 36(2), pp. 278-88.
- Westra, I., Oosterhuis, D., Groothuis, G. & Olinga, P., 2014. Precision-cut liver slices as a model for the early onset of liver fibrosis to test antifibrotic drugs. Toxicology and Applied Pharmacology, 274(2), pp. 328-38.
- White, D. L. et al., 2017. Incidence of hepatocellular carcinoma in all 50 United States, from 2000 through2012. Gastroenterology, 152(4), p. 812–820.e5.
- Wiegand, J. &Berg, T., 2013. The etiology, diagnosis and prevention of liver cirrhosis: Part 1 of a series on liver cirrhosis. Deutsches Arzteblatt International, 110(6), p. 85– 91.
- Winchester, C. L. & Salji, M., 2016. Writing a literature review. Journal of clinical urology,9(5), pp. 308-12.
- Wong, G. L. et al., 2014. Coincidental metabolic syndrome increases the risk of liver fibrosis progression in patients with chronic hepatitis B – A prospective cohort study with paired transient elastography examinations. Alimentary Pharmacology & Therapeutics, 39(8), pp. 883-93.
- Wong, V. et al., 2013. Community-based lifestyle modification programme for nonalcoholic fatty liver disease: A randomized controlled trial. Journal of Hepatology, 59(3), pp. 536-42.
- Wong, V. et al., 2016. Pathogenesis and novel treatment options for non-alcoholic steatohepatitis. Lancet Gastroenterology & Hepatology. 1(1), pp. 56-67.
- Woolbright, B. L., Ding, W. X. & Jaeschke, H., 2018. Caspase inhibitors for the treatment of liver disease: friend or foe. Expert Review of Gastroenterology & Hepatology, 11(5), p. 397–99.
- Yoon, H. et al., 2013. Effects of metabolic syndrome on fibrosis in chronic viral hepatitis. Gut and Liver, 7(4), p. 469–74.
- Younossi, Z., Stepanova, M. & Afendy, M., 2011. Changes in the prevalence of the most common causes of chronic liver diseases in the United States from 1988 to 2008. Clinical Gastroenterology and Hepatology, 9(6), pp. 524-530.e1.
- Zhang, Y. et al., 2018. Therapeutic effect of hepatocyte growth factor-overexpressing bone marrow-derived mesenchymal stem cells on CCl4-induced hepatocirrhosis. Cell Death & Disease, 9(12), p. 1186.
- Zhao, B. et al., 2016. Evaluation of the efficacy of steroid therapy on acute liver failure. Experimental and Therapeutic Medicine, 12(5), p. 3121–129.
- Zhou, J. et al., 2019. Combined obeticholic acid and apoptosis inhibitor treatment alleviates liver fibrosis. Acta Pharmaceutica Sinica B, 9(3), pp. 526-36.
- Zhou, W. C., Zhang, Q. B. & Qiao, L., 2014. Pathogenesis of liver cirrhosis. World Journal of Gastroenterology, 20(23), p. 7312–324.
- Zimmermann, H. W. et al., 2010. Functional contribution of elevated circulating and hepatic non-classical CD14CD16 monocytes to inflammation and human liver fibrosis. PloS One, 5(6), p. e11049.
© Copyright 2021, All Rights Reserved. Use of this content signifies your agreement to the T&Cs of Unified Citation Journals
This abstract of Manuscript/Paper/Article is an open access Manuscript/Paper/Article distributed under the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0/) which allows and permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited and accepted.
This communication and any documents, or files, attached to it, constitute an electronic communication within the scope of the Electronic Communication Privacy Act (https://it.ojp.gov/PrivacyLiberty/authorities/statutes/1285)
To citation of this article: Dr. Laila Alshammari, Development of Effective Therapies against Liver Fibrosis, Global Journal of Pathology & Laboratory Medicine